

ORIGINAL ARTICLE
A CHATBOT FOR IDENTIFYING AND MONITORING TOXICITIES ASSOCIATED WITH ANTINEOPLASTIC CHEMOTHERAPY IN PATIENTS WITH CANCER
Aline da Costa Novack1, Luciana Puchalski Kalinke2, Jacqueline Munhoes dos Santos Soares3, Larissa Marcondes4, Sonia Silva Marcon5, Luciana de Alcantara Nogueira6
1 Federal University of Paraná. Curitiba, PR, Brazil. ORCID: 0009-0006-0093-9984. E-mail: aline.novak@ufpr.br
2 Federal University of Paraná. Curitiba, PR, Brazil. ORCID: 0000-0003-4868-8193. E-mail: kalinkeluciana@gmail.com
3 Federal University of Paraná. Curitiba, PR, Brazil. ORCID: 0009-0002-6250-3562. E-mail: jacqueline.munhoes@ufpr.br
4 Federal University of Paraná. Curitiba, PR, Brazil. ORCID: 0000-0002-8745-6486. E-mail: marcondes.lari@gmail.com
5 State University of Maringá. Maringá, PR, Brazil. ORCID: 0000-0002-6607-362X. E-mail: soniasilva.marcon@gmail.com
6 Federal University of Paraná. Curitiba, PR, Brazil. ORCID: 0000-0002-5985-7418. E-mail: luciana.nogueira@ufpr.br
ABSTRACT
Objective: To develop a chatbot for the identification and monitoring of toxicities associated with intravenous antineoplastic chemotherapy in patients with cancer. Method: This methodological study was conducted between December 2023 and August 2025 and was based on the Interdisciplinary Method for the Development of Health Technologies, which comprises two phases and six activities. Phase 1 included Activity 1 – problem understanding and scoping review; Activity 2 – literature review; and Activity 3 – development of a low-fidelity prototype, prototype evaluation by nursing staff, and development of a high-fidelity prototype. Phase 2 comprised Activity 4 – development of the interactive prototype; Activity 5 – usability analysis and content validation; and Activity 6 – final writing and technology registration. Results were analyzed using the Content Validity Index (CVI > 90%) and usability assessment conducted with the System Usability Scale (score range, 0-100). Results: Nine symptoms most frequently reported in the literature supported the development of the chatbot. In the usability evaluation, 74.4% of users rated the system at the highest possible level of usability. In the content validation process, the achieved CVI was 90%. Conclusion: The chatbot demonstrated satisfactory content validity and usability, characterizing it as an appropriate technology to support health services in the remote monitoring of toxicities associated with chemotherapy, facilitating early identification and timely interventions.
Descriptors: Remote Patient Monitoring; Oncology; Drug-Related Side Effects and Adverse Reactions; Biomedical Technology.
|
Como citar: Novack AC, Kalinke LP, Soares JMS, Marcondes L, Marcon SS, Nogueira LA. A chatbot for identifying and monitoring toxicities associated with antineoplastic chemotherapy in patients with cancer. Online Braz J Nurs. 2025;24(Suppl 2):e20256946. http://doi.org/10.17665/1676-4285.20256946 |
What is already known:
Information and communication technologies are considered essential tools in contemporary clinical practice.
Monitoring toxicities related to intravenous antineoplastic chemotherapy can support health care teams in clinical decision-making.
What this article adds:
Chatbots represent a valid technological alternative for monitoring toxicities associated with intravenous antineoplastic chemotherapy.
The incorporation of a chatbot into oncology nursing practice has the potential to enhance care processes.
INTRODUCTION
The use of mobile technologies in health care, also referred to as mobile health (mHealth), is part of the broader field of eHealth and encompasses health-related tools that enable therapeutic processes, learning activities, and health promotion actions. According to the World Health Organization, both terms refer to the use of information and communication technology (ICT) resources to support health care. Within the eHealth context, mHealth is distinguished by the use of mobile and wireless technologies, such as smartphones, smartwatches, patient monitoring devices, and personal digital assistants, particularly mobile software applications, to support health-related actions(1-2).
ICTs are considered essential instruments in contemporary clinical practice because they enable rapid data collection and transmission, facilitate communication between health professionals and patients, and expand access to relevant information, thereby contributing to information delivery to target populations(3). By allowing home monitoring, real-time storage, and visualization of clinical information, these technologies make a substantial contribution to clinical decision-making and to the quality of care provided(4). Thus, ICTs encompass different forms of information transmission and include all technologies that influence communication processes(5).
Among the health technologies currently available, chatbots, also known as virtual assistants based on conversational systems, have gained prominence due to their ability to interact with users in an automated, personalized, and continuous manner. These are software applications that may rely on artificial intelligence or on preconfigured response rules, using conversational flows to decode written or spoken questions, query databases, and generate responses, thereby simulating human dialogue(6-7). Chatbots have been applied across different fields, including initiatives led by the Brazilian Ministry of Health, which, in 2023, adopted these tools in the context of vaccination to provide information on campaigns, target populations, dosing schedules, characteristics, benefits, and the importance of immunization as well as to address misinformation(8).
Chatbots offer advantages over traditional information services, as they clarify users’ questions, are associated with lower costs, provide immediate responses, and remain available regardless of time or day. In oncology, these technologies can contribute to improved care outcomes by expanding and facilitating access to educational actions related to cancer for patients and caregivers. In addition, they show high user acceptability and have demonstrated effectiveness in automating tasks related to cancer screening, treatment, symptom management, and survivorship(9).
With regard to symptom management, chatbots may serve as a supportive resource for both patients and health professionals. By receiving information and assisting patients in understanding their symptoms, these tools enable monitoring of symptom occurrence and severity. In this way, the technology can support clinical decision-making by the health care team, representing a relevant tool in the chemotherapy setting, in which adverse events are frequent.
Specifically in the context of antineoplastic chemotherapy, despite advances in therapeutic processes, adverse events resulting from the use of antineoplastic agents remain a major challenge for health care teams. Although treatment increasingly represents an opportunity for cure or disease stabilization for patients, associated toxicities may compromise quality of life, affect treatment adherence, and require timely clinical interventions(10).
In this regard, the identification of toxicities is essential, as it informs decisions such as dose adjustments and the continuation or discontinuation of the prescribed treatment(11). However, home-based monitoring of symptoms remains incipient(12).
The present study is justified by the fact that the research setting is a reference oncology hospital in southern Brazil that lacks systematized methods for monitoring toxicities associated with chemotherapy. Therefore, the proposed approach represents an alternative with the potential to reduce the severity of in-person visits related to treatment toxicities, thereby contributing to greater patient safety.
Accordingly, the objective of this study was to develop a chatbot for the identification and monitoring of toxicities associated with intravenous antineoplastic chemotherapy in patients with cancer.
METHOD
This was a methodological study conducted at a reference hospital for cancer treatment in southern Brazil. The Interdisciplinary Method for the Development of Health Technologies (IMDHT) was adopted. This method comprises two phases and six activities, as follows: Phase 1 – Activity 1: problem identification, scoping review, and definition of the guiding research question; Activity 2: literature review; Activity 3: development of a low-fidelity prototype, evaluation of the prototypes by expert nurses, and development of high-fidelity prototypes. Phase 2 – Activity 4: development of the interactive prototype; Activity 5: usability analysis and content validation; and Activity 6: product registration.
Phase 1
Phase 1 – Activity 1 was initiated based on the identification of the absence of monitoring of toxicities associated with antineoplastic chemotherapy in the study setting, which constituted a service-related problem. After defining the problem, a scoping review was conducted(13-14) and published in Revista Ciência, Cuidado e Saúde (DOI: 10.4025/ciencuidsaude.v24i1.72599), with the aim of mapping scientific evidence on remote monitoring of toxicities in adult patients with cancer undergoing chemotherapy. The protocol for this scoping review was registered on the international Open Science Framework platform (DOI: 10.17605/OSF.IO/BK7ZF).
To formulate the guiding research question, the PCC mnemonic framework was used, in which P (population) referred to adult patients with cancer; C (concept) to remote monitoring; and C (context) to antineoplastic chemotherapy. Based on these definitions, the guiding question was defined as follows: What do high-impact studies report regarding the remote monitoring of adult patients with cancer undergoing chemotherapy?
Search was conducted in the PubMed, Web of Science, Cumulative Index to Nursing and Allied Health Literature (CINAHL), Embase, and Virtual Health Library (BVS) databases. Primary studies published in open access, in any language and without time restriction, were included. During the eligibility stage, only clinical trials, whether randomized or nonrandomized, were included to ensure a high level of evidence. Study design classification was based on the terminology used by the authors themselves.
The findings of this scoping review guided the researchers toward the development of a chatbot, as it represented a less costly, practical, and feasible alternative for the investigated context. In light of the identified problem and with the intention of developing a technology for remote monitoring, the following guiding question was established for the study: Can a chatbot be a valid tool for monitoring toxicities caused by intravenous chemotherapy?
Activity 2 aimed to identify the content to be incorporated into the chatbot. A literature review was conducted to identify the main toxicities and symptoms associated with intravenous antineoplastic chemotherapy.
Literature review was carried out using the Portal of Journals of the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Ministry of Education, with searches in the Embase, Web of Science, Scopus, and BVS databases. The PCC mnemonic was again applied, with P (population) referring to adult patients with cancer; C (concept) to toxicity and adverse events; and C (context) to antineoplastic chemotherapy. Open-access articles published between 2019 and March 2024, in Portuguese, English, or Spanish, were included if they contained the keywords side effects, adverse drug reactions, toxicity, antineoplastic agents, and neoplasms. The search strategy used in the Embase and Scopus databases was (chemotherapy AND “drug toxicity” AND “adverse event”). In Web of Science, the strategy was (chemotherapy AND “drug toxicity”), and in the BVS portal, (“quimioterapia e toxicidade”).
Activity 3 consisted of developing a low-fidelity interface prototype, defined as a preliminary drawing or sketch of the technological product to be developed(15). This activity was divided into two stages: I) development of the low-fidelity chatbot interface prototype and II) evaluation of the low-fidelity prototype by nurse evaluators.
During the development of low-fidelity prototypes, the identified symptoms were designated as Access Menus. Dialogue scripts for each menu (symptom) were developed using Microsoft Word, with prototypes created in a panel format representing interaction screens or dialogues organized sequentially.
Nine Access Menus were developed: Initial Access Menu (screens 1-5); Nausea Access Menu; Vomiting Access Menu; Diarrhea Access Menu; Constipation Access Menu; Tingling Sensation in Hands and Feet/Peripheral Neuropathy Access Menu; Fatigue/Tiredness Access Menu; Loss of Appetite/Inappetence Access Menu; and Phlebitis/Vein Swelling Access Menu.
Each menu panel contained a sequence of screens representing the order of the conversation, an animation indicating the expected chatbot behavior, the chatbot content and/or interaction expressing the software’s communication style, language, and expressions, the user interaction corresponding to responses provided to the chatbot’s questions, and a column dedicated to alerts and managerial data. This latter column included response options to be registered by the programming team as notification priorities, as they indicated signs of clinical worsening according to the Common Terminology Criteria for Adverse Events (CTCAE 4.0), as defined by the researcher.
CTCAE 4.0 is a descriptive terminology document designed to assess the severity of toxicities on a scale from 0 to 5, aiming to standardize and harmonize toxicities resulting from oncological treatment, as described by the National Cancer Institute(16).
The Initial Access Menu was common to all symptoms and comprised screens 1-5, which provided guidance on device operation and collected user data, such as name, national identification number, and date of birth. After this stage, participants selected the desired symptom, and subsequent screens referred specifically to that symptom, following the same numerical sequence.
Evaluation of the low-fidelity prototype was performed by 15 nurses, referred to as evaluators. All nurses working in the institution’s infusion centers were invited to a meeting in which the principal investigator presented the study objectives and obtained written informed consent from those who agreed to participate. The informed consent form addressed aspects related to the Brazilian Data Protection Law, ensuring participants’ right to data confidentiality, and was developed in accordance with the criteria of the institution’s Research Ethics Committee, which approved the project.
Inclusion criteria for participation in this activity were having more than 6 months of experience in direct care of oncology patients. Nurses on leave or working exclusively in managerial roles were excluded. To evaluate the prototypes, a 5-point Likert-scale questionnaire was developed and analyzed using the content validity index (CVI), addressing aspects related to objectives, design concept, and the sequence of presentation of each prototype.
High-fidelity prototypes were developed using the Canva design tool to improve dialogue visualization and make them more similar to the final technology. Canva was used exclusively for mockup creation.
Phase 2
Phase 2 – Activity 4 began with the creation of specific dialogue screens corresponding to the content of each Access Menu. Thus, when reporting a given symptom, such as nausea, users were automatically directed to exclusive screens related to that symptom. This stage involved information technology (IT) professionals responsible for creating the interaction flows within the WhatsApp application. To ensure patient data security, the chatbot was not integrated with the hospital’s operational system. Generated notifications were sent to a dashboard configured as a web page with restricted access via username and password. The programming language used by the IT team was not disclosed.
Activity 5 was conducted in two stages: i) usability analysis of the chatbot, performed by patient participants; and ii) content validation, performed by nurse evaluators.
For the usability analysis, the chatbot was made available to 47 adult patients undergoing intravenous chemotherapy at the study hospital. Participants were approached in person by the researcher, who explained the study objectives, research design, and the functioning of the tool for home use. Each patient received 10-15 minutes of guidance on chatbot use. All invited patients agreed to participate, with no sample losses.
Inclusion criteria for the usability analysis were age ≥ 18 years; receipt of intravenous antineoplastic chemotherapy; ownership of a mobile phone with internet access; and weekly or biweekly chemotherapy cycles. Patients who reported difficulties using mobile devices were excluded.
The usability sample was defined by convenience, in accordance with the principles of the IMDHT, which does not establish a minimum number of participants. Recruitment therefore occurred over 1 week, during which all patients receiving intravenous chemotherapy were approached, with no refusals. For content validation, the recommended range of 5-20 experts was adopted(17).
After participants’ understanding and agreement, written informed consent was obtained. At this time, the researcher assisted participants in saving the chatbot’s WhatsApp contact on their mobile phones and supervised the first interaction, clarifying any questions. Patients used the chatbot for a mean period of 20 days and, at the end of this interval, were contacted again to complete the System Usability Scale (SUS), translated and adapted for Brazilian Portuguese(18).
Content validation of the chatbot prototype was performed using a specific questionnaire designed to assess aspects related to objectives, language, expressions, texts, and data presented by the technology. Inclusion criteria for this stage were at least 6 months of experience in direct care of oncology patients. Nurses holding other positions or on leave for any reason were excluded. No additional exclusion criteria were applied.
Activity 6 comprised preparation of the final research manuscript and registration of copyright, under the transaction hash: 0xca5bc7482747d5ea80b84d50153e3c718dd5eaeef97e56b747a4662b95cb7e00.
For analysis of data from the evaluation conducted in Phase 1 and from content validation in Phase 2, CVI was calculated by summing responses rated as 4 or 5 and dividing by the total number of responses. CVI expresses the percentage of agreement among evaluators for each item and is considered satisfactory when > 90%(17).
In Phase 2, usability results were analyzed according to SUS recommendations. For odd-numbered items (1, 3, 5, 7, and 9), 1 point was subtracted from the score assigned by the respondent; for even-numbered items (2, 4, 6, 8, and 10), the assigned score was subtracted from 5. The resulting values were summed and multiplied by 2.5(19).
Final SUS scores range from 0 to 100 and represent a usability metric, classified as follows: ≤ 25 points, worst possible usability; > 25 to < 40 points, poor usability; 40-52 points, fair usability with potential issues; 53-73 points, good usability; 74-85 points, excellent usability; and ≥ 85 points, best possible usability(19).
Because of the extent of the low-fidelity, high-fidelity, and interactive prototypes, only selected interactions are presented in the Results section. The complete material is currently being made available in an electronic repository. Regarding ethical aspects, the study was reviewed and approved by the Human Research Ethics Committee (opinion No. 6.915.260). All participants provided written informed consent.
RESULTS
The study results are presented according to the respective phases.
Phase 1
The scoping review initially identified 2,611 publications. After duplicate removal and application of the inclusion criteria, six studies comprised the final sample.
The selected studies aimed to evaluate and test the effectiveness and feasibility of remote monitoring. Results showed variability in outcomes related to the mean number of emergency department visits, hospitalizations, and use of supportive care. The modalities employed for remote monitoring included mobile applications, telephone calls, text messages, and telephone-based systems. The scoping review concluded that remote monitoring of adult patients with cancer undergoing chemotherapy has the potential to positively influence the period between chemotherapy cycles by providing support to both patients and health care teams. These findings guided the development of a chatbot, as it represents a less costly, practical, and feasible alternative for the investigated context. Thus, the research team initiated a literature review to identify the most frequent symptoms in patients undergoing chemotherapy in order to inform the chatbot content.
The literature review identified 1,862 articles and, after application of the inclusion criteria, 19 studies were selected for full-text review. Analysis of these studies identified 14 symptoms commonly reported among patients undergoing chemotherapy. However, symptoms cited only once in the included studies were not incorporated into the chatbot. Thus, the most recurrent symptoms that supported the development of the low-fidelity prototype were nausea, vomiting, diarrhea, constipation, peripheral neuropathy, weakness/fatigue, loss of appetite, phlebitis, and injection-site pain. Chart 1 presents an example of the Nausea Access Menu.
Chart 1 – Low-fidelity chatbot prototype for the Nausea Access Menu. Curitiba, PR, Brazil, 2025
|
Screen |
Animation |
Chatbot content/interaction |
User interaction |
Alerts and managerial data |
|
Screen 06 |
Explanation of the selected symptom |
“Nausea is an unpleasant feeling in the stomach, often accompanied by the urge to vomit. What an uncomfortable sensation!”
“Is this what you are experiencing?” 01 – Yes, proceed 02 – No, return to the initial menu |
If response 01, proceed with the Nausea Access Menu interaction. If response 02, return to Screen 05 of the Initial Access Menu. |
Identification of symptom severity using CTCAE 4.0. |
|
|
|
“I see! Tell me how nausea is affecting your eating:” 01 – Loss of appetite, without changes in food intake; 02 – Reduced oral intake, with significant weight loss; 03 – Little or no oral intake of food or fluids. |
Entry according to symptom presentation (e.g., 01). |
If response 01 – access recorded in the management dashboard, with no alert issued. |
|
|
|
If response 01: “I noticed that your symptoms are at an early stage. You should continue monitoring and use the anti-nausea medications prescribed by your physician at the initial or follow-up visit.” “If there are any changes, please submit a new report.” |
|
|
|
|
|
If response 02 or 03: “Hmm, I understand, you are not feeling well!” “I am sending a notification to the nursing team, and you may soon receive a phone call from one of our nurses.” “Most importantly, please seek emergency care at the hospital as soon as possible for medical evaluation.” |
|
If response 02 or 03 – email notification sent and alert generated in the management dashboard. |
Source: prepared by the authors, 2025.
The evaluation of the low-fidelity prototypes, conducted by 15 nurses, yielded a CVI of 93%. As this value exceeds 90%, the result was classified as satisfactory. Therefore, no adjustments to the dialogue interactions of the low-fidelity prototypes were required.
The high-fidelity prototypes enabled a clearer understanding of the final product, as they simulated a conversation within the WhatsApp application and allowed visualization of the dialogue boxes exchanged between the chatbot and the user. Figure 1 presents the screens of the high-fidelity prototype.
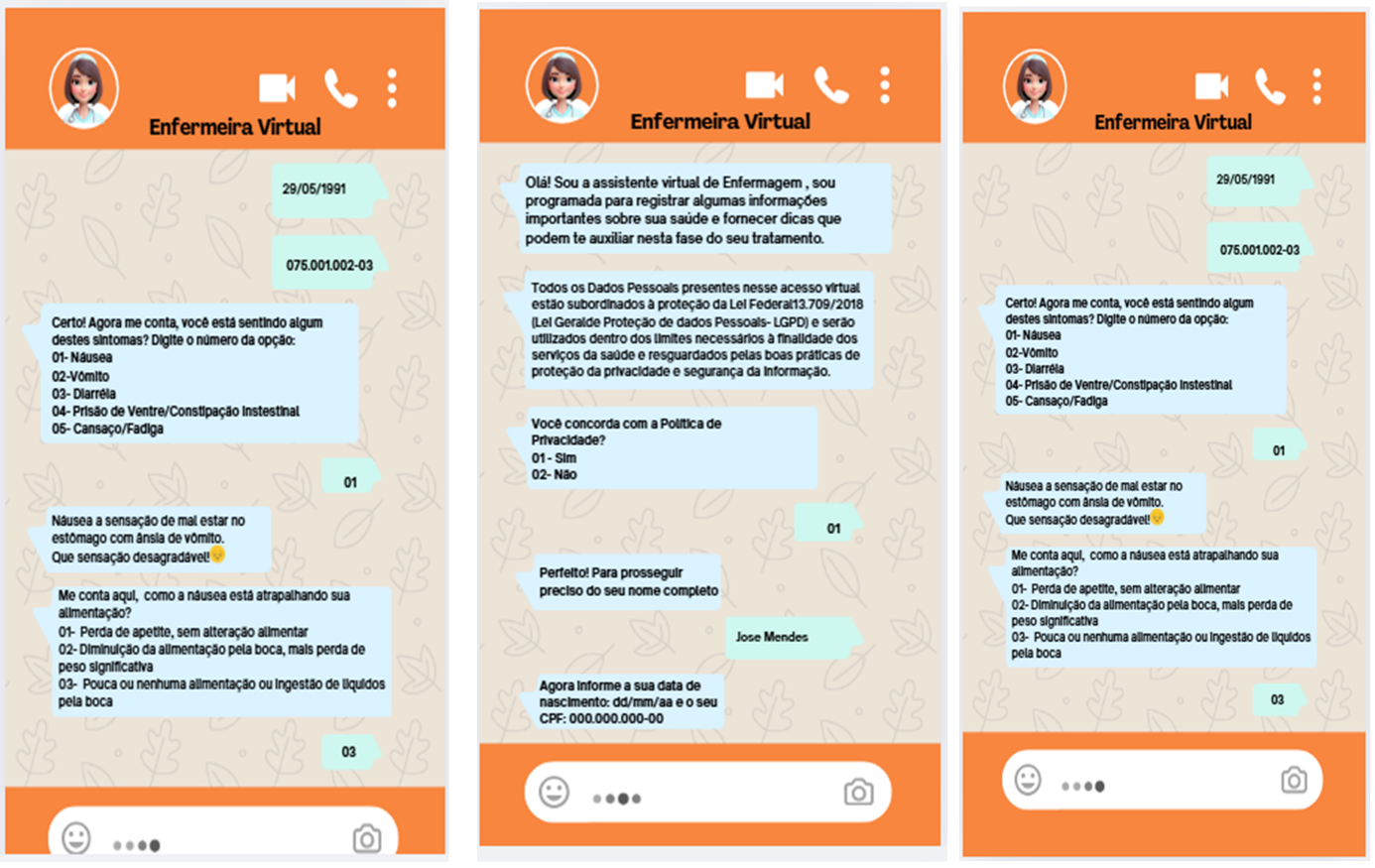
Source: prepared by the authors, 2025.
Figure 1 – High-fidelity chatbot prototype. Curitiba, PR, Brazil, 2025
Phase 2
After completion of Phase 1, the high-fidelity prototypes were submitted to the IT team for development and programming of the interactive prototypes. Figure 2 illustrates part of the real interaction of the developed interactive prototypes.
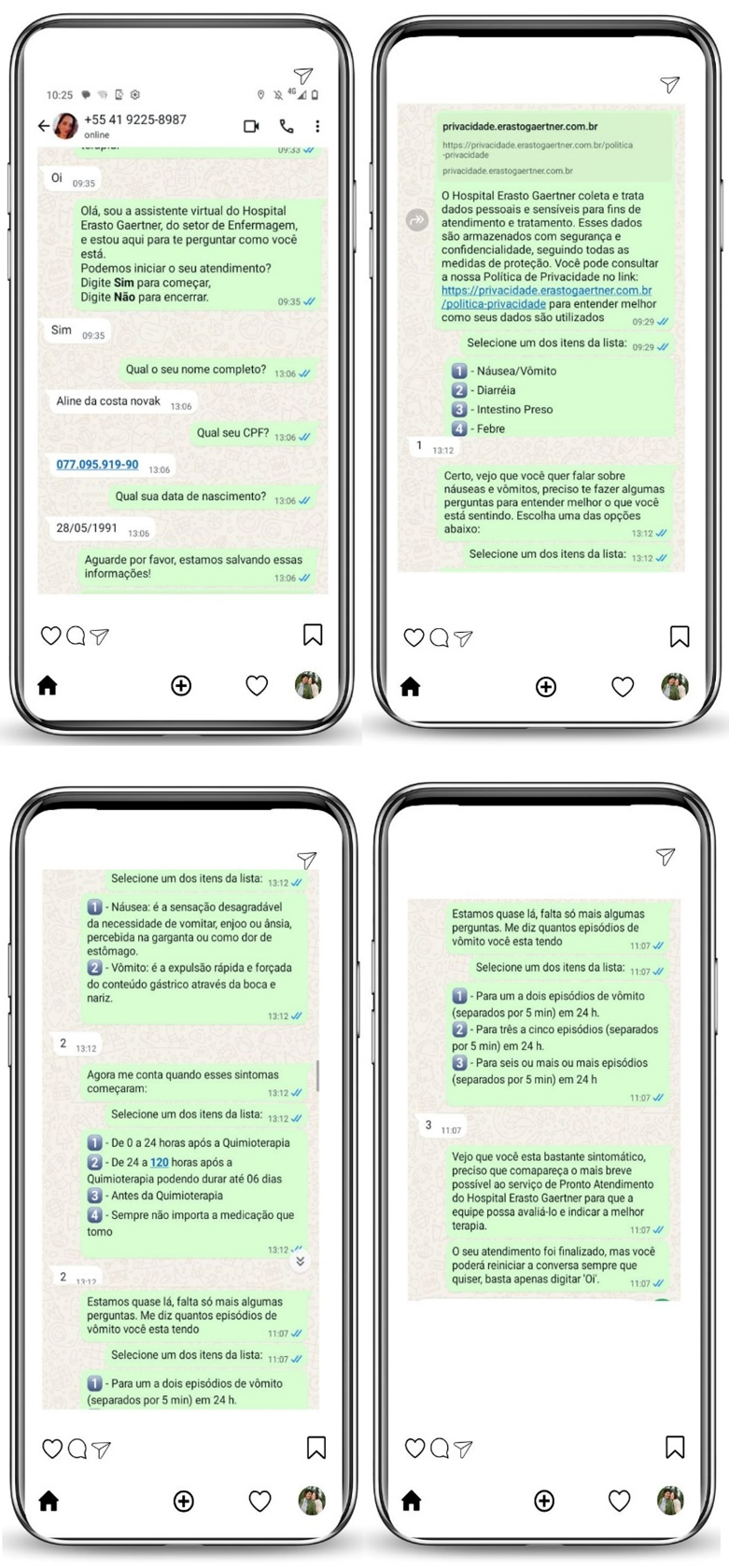
Source: prepared by the authors, 2025.
Figure 2 – Interactive chatbot prototype. Curitiba, PR, Brazil, 2025
Regarding the characteristics of the 47 patients who participated in the usability analysis, 55.3% were female, with a mean age of 60 years. In terms of educational level, 44.6% of the sample had completed high school and 25.5% had completed primary education. With respect to diagnosis, 64% of participants had gastrointestinal cancer and 19.1% had breast cancer.
Concerning chatbot usability, 35 patients (74.4%) rated the system as having the best possible usability, 10 (21.7%) rated it as excellent usability, and 2 (4.2%) rated it as good usability. According to SUS criteria, the chatbot’s usability was classified as the best possible, indicating that most participants were able to use the technology easily, without the need for adjustments, corrections, or additional support.
The fact that the chatbot operated through a conversational interface within the WhatsApp application, a tool widely integrated into daily life, particularly after the COVID-19 pandemic, facilitated use of the technology, with no reported dropouts or difficulties. Consequently, all participants completed toxicity reporting during the home-use period.
Regarding content validation, the 15 nurses accessed the chatbot system to interact with the conversational agent and, after completing the content validation questionnaire, the chatbot achieved a CVI of 90% (Table 1).
Table 1 – Content validation of the chatbot by nurse evaluators (n = 15). Curitiba, PR, Brazil, 2025
|
Chatbot content validation items |
No. of responses = 4 |
No. of responses = 5 |
CVI |
|
1) The chatbot objectives are consistent with the proposed symptom monitoring. |
1 |
13 |
0.93 |
|
2) The chatbot facilitates users’ understanding of their symptoms. |
2 |
13 |
1.00 |
|
3) The proposed objectives are appropriate for their implementation. |
10 |
5 |
1.00 |
|
4) The content title represents its objectives. |
2 |
12 |
0.93 |
|
5) The chatbot content corresponds to the symptom and its description. |
1 |
14 |
1.00 |
|
6) The information presented is well structured. |
2 |
13 |
1.00 |
|
7) The content facilitates learning regarding chemotherapy-related symptoms. |
2 |
13 |
1.00 |
|
8) The name of each stage/phase is consistent with the content presented. |
2 |
13 |
1.00 |
|
9) The operational steps are well described for execution of the stages/phases. |
1 |
14 |
1.00 |
|
10) There is a logical sequence of ideas and their stages/phases. |
2 |
13 |
1.00 |
|
11) The model information is clear, objective, and representative of the proposal. |
2 |
13 |
1.00 |
|
12) The vocabulary used is understandable to respondents. |
2 |
13 |
1.00 |
|
Mean of validated items (S-CVI) |
|
|
0.90 |
Source: prepared by the authors, 2025.
Complementing the results, during the period in which the chatbot was available to the 47 patients, a total of 142 accesses were recorded. All participants accessed the system, with a mean of 3 accesses per patient. The first access generally occurred between the 3rd and 7th day after the start of use. Figure 3 shows the percentage distribution of accesses according to the symptom reported.
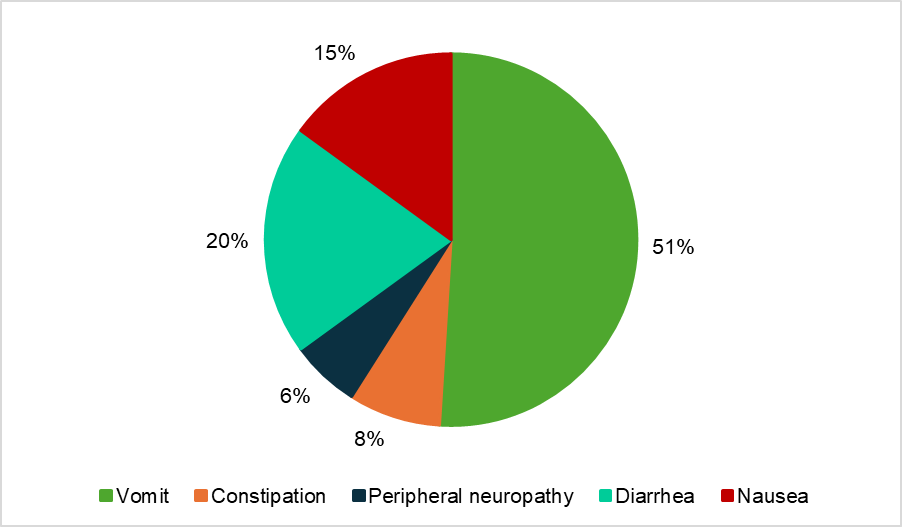
Source: prepared by the authors, 2025.
Figure 3 – Symptoms accessed in the chatbot during the usability analysis (n = 47). Curitiba, PR, Brazil, 2025
DISCUSSION
This study demonstrated that chatbots constitute a valid technological alternative for monitoring toxicities associated with intravenous antineoplastic chemotherapy. Research on the use of chatbots in health treatments or user experience remains limited and continues to represent an innovative initiative within oncology nursing, which underscores the relevance of the present study(20).
Chatbots stand out for their usability, accessibility, and ability to disseminate information rapidly and in an automated manner, optimizing human work and enabling the expansion of health care delivery. These tools enhance human capacities for care provision and user communication, while also automating simple and repetitive tasks, allowing simultaneous responses to multiple demands, particularly because they are accessible through mobile devices(4).
According to Resende et al.(21), the widespread adoption of smartphones represents a new milestone in the relationship between society and information and communication technologies, as it integrates technology into social practices and enables the development of new forms of interaction with different linguistic logics and articulations. In this context, chatbots emerge as strategic tools for the storage, processing, and sharing of clinical data among health professionals, supporting faster and more accurate decision-making(22).
It is thus essential for nurses to adapt to technological advances by seeking continuous professional qualification and improvement. As central agents of comprehensive care, nurses must be prepared to use these resources ethically and efficiently, contributing to improved health indicators and patients’ quality of life(23).
In oncology, a study conducted in 2019 demonstrated a survival gain of up to 9 months among a group of 133 patients monitored remotely, suggesting that early detection of symptoms related to lung cancer progression enabled immediate interventions and reduced negative impacts. These findings also highlight greater patient engagement in care and emphasize the importance of considering the patient perspective and the impact of treatment on daily life(24).
Findings from the first phase of this study, based on the scoping review, indicated that the chatbot represented a feasible and innovative solution to the problem identified in the oncology service. However, the literature highlights the need for studies assessing the usability impact of these technologies among target populations, particularly regarding compatibility with users’ cognitive levels and barriers to digital adherence, as many studies focus primarily on technology development and implementation(25).
The literature review that supported chatbot development identified prevalent symptoms such as nausea and vomiting, corroborating the findings of Marinho et al.(26), who reported that among 140 women with breast cancer, 84.3% experienced nausea and 90.7% experienced vomiting during treatment. These results were also confirmed in the usability analysis phase of the present study, in which vomiting accounted for more than half of chatbot accesses.
Usability analysis and content validation conducted with users and professionals indicated that the technology has the potential to meet service demands for remote monitoring of patients undergoing intravenous chemotherapy, even considering a sample predominantly composed of older adults. This finding is consistent with the study by Denis et al.(24), which assessed the feasibility of smartphone use for cognitive capacity evaluation in 57 older adults and demonstrated that home-based application of these technologies is feasible, comfortable, and cost-effective.
Morris et al.(27) developed an empathetic conversational agent designed to reproduce human-like responses, which was evaluated over a 2-week period by 37,169 users with anxiety and depression, showing a significant reduction in reported symptoms. This finding suggests that usability and evaluation studies can be conducted over relatively short periods without compromising result quality.
Regarding the accesses recorded during the usability analysis, older users may experience greater difficulties in using the technology. However, recent data from the Brazilian Institute of Geography and Statistics(28) show a substantial increase in internet access among older adults in Brazil, rising from 24.7% in 2016 to 62.1% in 2022. This trend suggests growing digital inclusion and reinforces the potential of tools such as chatbots for this population.
With respect to content validation performed by expert nurses, the high level of agreement among evaluators indicates the adequacy of the structural, communicational, and informational elements of the proposed technology.
Study limitations include the scarcity of similar research in the field, which restricted comparative discussion; the short period of chatbot use by participants, limiting assessment of long-term engagement and sustainability throughout treatment; conduction of the study at a single center, which may limit generalizability; use of a convenience sample in the usability analysis; and the absence of evaluation of health care team satisfaction with system-generated alerts.
CONCLUSION
Execution of the study stages resulted in the development of a technology with satisfactory content validity (CVI of 90%) for the remote monitoring of toxicities associated with intravenous chemotherapy. The chatbot proved capable of registering toxicities in a structured manner and of timely signaling the health service, facilitating early interventions and adjustments to the therapeutic plan.
Incorporation of the chatbot into oncology nursing practice has the potential to enhance care processes by enabling continuous patient follow-up during treatment and improving communication between users and health care teams.
*Article extracted from the Master's Dissertation entitled “Technology in health: development of a chatbot for monitoring adverse events associated with intravenous chemotherapy in adult cancer patients”, presented to the Postgraduate Program: Practice of Health Care, Federal University of Paraná, Curitiba, PR, Brazil, in 2025.
CONFLICT OF INTEREST
The authors declare no conflict of interest.
REFERENCES
1. Valerio Netto A, Salvador ME. Challenges associated with the development of scientific projects and studies in digital health and mobile technologies. Rev Bras Enferm. 2020;73(6):e73n6. https://doi.org/10.1590/0034-7167.202073n601
2. Marengo LL, Kozyreff AM, Moraes F da S, Maricato LIG, Barberato-Filho S. Tecnologias móveis em saúde: reflexões sobre desenvolvimento, aplicações, legislação e ética. Rev Panam Salud Publica. 2023;46:e37. https://doi.org/10.26633/RPSP.2022.37
3. Silva NV de N da, Pontes CM, Sousa NFC de, Vasconcelos MGL de. Tecnologias em saúde e suas contribuições para a promoção do aleitamento materno: revisão integrativa da literatura. Cien Saude Colet. 2019;24(2):589-602. https://doi.org/10.1590/1413-81232018242.03022017
4. Scaratti M, Argenta C, Almeida WX de, Zanatta EA. Mobile application resources to selfcare and selfmanagement of type i diabetes mellitus: integrative review. Rev. Pesq.: Cuid. Fundam. 2021;13:1374-1380. https://doi.org/10.9789/2175-5361.rpcfo.v13.10020
5. Rodrigues FMS, Hinds PS, Nascimento LC. Adolescents’ understanding of chemotherapy-related adverse events: a concept elicitation study. Rev Lat Am Enfermagem. 2022;30(spe):e3717. https://doi.org/10.1590/1518-8345.6245.3718
6. Lugli VA, Lucca Filho J de. O uso do chatbot para a excelência em atendimento. Interface Tecnol. (Online). 2020;17(1):205-218. https://doi.org/10.31510/infa.v17i1.840
7. Albuquerque M dos S, Silva CDS, Gomes DM, Pinto MMM, Vasconcelos SP de. SUSi: chatbot na promoção do conhecimento da atenção primária à saúde. Cad. ESP (Online). 2023;17(1):e1516. https://doi.org/10.54620/cadesp.v17i1.1516
8. Empresa Brasileira de Serviços Hospitalares. Câncer: diagnóstico precoce aumenta chances de cura [Internet]. Brasília: Ministério da Saúde; 2023 [cited 2024 Feb 02]. Available from: https://www.gov.br/ebserh/pt-br/comunicacao/noticias/cancer-diagnostico-precoce-aumenta-chances-de-cura
9. Belfin RV, Shobana AJ, Manilal M, Mathew AA, Babu B. A Graph Based Chatbot for Cancer Patients. Proceedings of the 2019 5th International Conference on Advanced Computing & Communication Systems; 2019 Mar 15-16; Coimbatore, India: IEEE; 2019. p. 717-721. https://doi.org/10.1109/ICACCS.2019.8728499
10. Caldeira BM, Galbiatti-Dias ALS. Acompanhamento e avaliação dos efeitos adversos da quimioterapia em pacientes com câncer de mama. Revista Corpus Hippocraticum [Internet]. 2017 [cited 2024 Apr 18];1(1). Available from: https://revistas.unilago.edu.br/index.php/revista-medicina/article/view/8
11. Iuchno CW, Carvalho GPD. Toxicidade e efeitos adversos decorrente do tratamento quimioterápico antineoplásico em pacientes pediátricos: revisão integrativa. Ciênc. Saúde (Porto Alegre). 2019;12(1):e30329. https://doi.org/10.15448/1983-652X.2019.1.30329
12. Escobar GF. Incorporação de soluções digitais em oncologia e seu impacto na experiência do paciente [master's thesis]. São Paulo: Fundação Getulio Vargas; 2021.
13. Aromataris E, Lockwood C, Porritt K, Pilla B, Jordan Z, editors. JBI Manual for Evidence Synthesis. Adelaide: JBI; 2024. https://doi.org/10.46658/JBIMES-24-01
14. Tricco AC, Lillie E, Zarin W, O’Brien KK, Colquhoun H, Levac D, et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): Checklist and Explanation. Ann Intern Med. 2018;169(7):467-473. https://doi.org/10.7326/m18-0850
15. Vasconcelos Filho JE, Brasil CCP, Rolim KMC, Silva Junior GB da, Silva RM da. MIDTS: método interdisciplinar para o desenvolvimento de tecnologias em saúde. In: Jorge MSB, Vergara CMAC, Sampaio HA de C, Moreira TMM, organizers. Tecnologias e-Health em Gestão em Saúde: fundamentos para seu desenvolvimento e avaliação. Curitiba: CRV; 2021. p. 49-66.
16. Tavares MB, Oliveira JR de, Silva VF da, Junior GB da S, Bendicho MT, Xavier RMF. Caracterização das reações adversas a quimioterápicos em um hospital filantrópico. Braz. J. Health Rev. 2020;3(2):2317-2326. https://doi.org/10.34119/bjhrv3n2-083
17. Alexandre NMC, Coluci MZO. Validade de conteúdo nos processos de construção e adaptação de instrumentos de medidas. Cien Saude Colet. 2011;16(7):3061-3068. https://doi.org/10.1590/S1413-81232011000800006
18. Lourenço DF, Carmona EV, Lopes MHB de M. Translation and Cross-Cultural Adaptation of the System Usability Scale to Brazilian Portuguese. Aquichan. 2022;22(2):e2228. https://doi.org/10.5294/aqui.2022.22.2.8
19. Majer CA, Duduchi M. Avaliação de usabilidade de simulador brasileiro de jogo de empresas. Braz J Dev. 2019;5(11):23768-23777. https://doi.org/10.34117/bjdv5n11-078
20. Roque G da SL, Souza RR de, Nascimento JWA do, Campos Filho AS de, Queiroz SR de M, Santos ICRV. Content validation and usability of a chatbot of guidelines for wound dressing. Int J Med Inform. 2021;151:104473. https://doi.org/10.1016/j.ijmedinf.2021.104473
21. Resende JVM, Silva JLL da, Soares R da S, Miranda P da S, Costa F dos S, Abreu WO de, et al. Aplicativos para celular na área da saúde: uma revisão integrativa. Res Soc Dev. 2022;11(11):e278111133481. https://doi.org/10.33448/rsd-v11i11.33481
22. Lima CSP, Barbosa S de FF. Mobile health applications: characterization of the scientific production of Brazilian nursing. Rev. Eletrônica Enferm. 2019;21:53278. https://doi.org/10.5216/ree.v21.53278
23. Garces KGV, Sousa TDV, Serra RB, Silveira WJA, Oliveira CA de S, Lima JG, et al. Saúde e bem-estar digital: uso de tecnologias na assistência de enfermagem. Europub Journal of Health Research. 2025;6(1):e5697. https://doi.org/10.54747/ejhrv6n1-006
24. Denis F, Basch E, Septans AL, Bennouna J, Urban T, Dueck AC, et al. Two-Year Survival Comparing Web-Based Symptom Monitoring vs Routine Surveillance Following Treatment for Lung Cancer. JAMA. 2019;321(3):306-307. https://doi.org/10.1001/jama.2018.18085
25. Ferreira MN, Ramseier JY, Leventhal JS. Dermatologic conditions in women receiving systemic cancer therapy. Int J Womens Dermatol. 2019;5(5):285-307. https://doi.org/10.1016/j.ijwd.2019.10.003
26. Marinho PML, Barbosa-Lima R, Santos JCDO, Santos DKDC, Costa JDS, Silva GM, et al. Toxicidades sistêmicas simultâneas relacionadas à quimioterapia do câncer de mama: um relato observacional e prospectivo. Rev. Enferm. Atual In Derme. 2022;96(40):e-021311. https://doi.org/10.31011/reaid-2022-v.96-n.40-art.1497
27. Morris RR, Kouddous K, Kshirsagar R, Schueller SM. Towards an Artificially Empathic Conversational Agent for Mental Health Applications: System Design and User Perceptions. J Med Internet Res. 2018;20(6):e10148. https://doi.org/10.2196/10148
28. Instituto Brasileiro de Geografia e Estatística. Pesquisa Nacional por Amostra de Domicílios Contínua: acesso à Internet e à televisão e posse de telefone móvel celular para uso pessoal 2023 [Internet]. Rio de Janeiro: IBGE; 2024 [cited 2025 Jul 29]. Available from: https://biblioteca.ibge.gov.br/visualizacao/livros/liv102107_informativo.pdf
Submission: 02-Sep-2025
Editors:
Rosimere Ferreira Santana (ORCID: 0000-0002-4593-3715)
Geilsa Soraia Cavalcanti Valente (ORCID: 0000-0003-4488-4912)
Corresponding author: Luciana de Alcantara Nogueira (luciana.nogueira@ufpr.br)
Publisher:
Escola de Enfermagem Aurora de Afonso Costa – UFF
Rua Dr. Celestino, 74 – Centro, CEP: 24020-091 – Niterói, RJ, Brazil
Journal email: objn.cme@id.uff.br
