

REVIEW PROTOCOL
COVID-19 IN INDIGENOUS POPULATIONS: A SCOPING REVIEW PROTOCOL
Priscila Carminati Siqueira1, Carolina Maia Martins Sales1, Jonathan Grassi Rodrigues1, Izabella Dossi Banhos1, Ethel Leonor Noia Maciel1
1 Federal University of Espírito Santo, ES, Brazil
ABSTRACT
Objective: To map the available evidence on COVID-19 in Indigenous populations. Method: This scoping review protocol was developed in accordance with the Scoping Reviews and Preferred Reporting Items for Systematic Reviews and Meta-Analyses Extension for Scoping Reviews (PRISMA-ScR) guidelines, following the Joanna Briggs Institute (JBI) methodology. Data sources will include MEDLINE/PubMed, Cochrane Library, Embase, Web of Science, Scopus, and LILACS as well as trial registries such as ClinicalTrials.gov and the WHO International Clinical Trials Registry Platform (ICTRP). Additional sources such as the British Library, Google Scholar, and Preprints for Health Sciences will also be searched, with no restrictions on date or language. Two independent reviewers will conduct the study selection, data mapping, and synthesis. The results will be presented through narrative synthesis, tables, and graphs.
Descriptors: COVID-19; Indigenous Peoples; Indigenous Population Health; Pandemics.
|
How to cite: Siqueira PC, Sales CMM, Rodrigues JG, Banhos ID, Maciel ELN. COVID-19 in Indigenous populations: a scoping review protocol. Online Braz J Nurs. 2025;24(Suppl 1):e20256825. https://doi.org/10.17665/1676-4285.20256825 |
INTRODUCTION
The infection caused by the SARS-CoV-2 virus was first identified in December 2019 in Wuhan, China, and is responsible for causing severe respiratory illnesses. On January 30, 2020, the World Health Organization (WHO) declared the outbreak of a public health emergency of international concern. Subsequently, on March 11, 2020, COVID-19 was characterized as a pandemic, with outbreaks reported in multiple countries and regions around the world(1).
More than 775 million confirmed cases of COVID-19 and over 7 million deaths have been reported worldwide(2). However, significant gaps remain in the literature regarding Indigenous populations affected by COVID-19, particularly with respect to the scarcity of demographic and epidemiological data. This lack of information limits the ability of authorities to interpret and understand the impact of COVID-19 on Indigenous communities, hindering timely decision-making to mitigate the effects of the pandemic in these territories(3).
The COVID-19 pandemic poses a major concern for Indigenous populations, as infectious and parasitic diseases remain among the leading causes of death in this group, particularly when compared to other ethnic and racial groups(3-10).
Throughout history, various epidemics of infectious and parasitic diseases have led to significant demographic declines among Indigenous peoples. Notable examples include the Spanish flu (1918–1919) and the H1N1 pandemic in 2009. During the latter, Indigenous populations experienced severe acute respiratory infections at a rate 4.5 times higher than that of other populations. These public health crises have consistently demonstrated the increased vulnerability of Indigenous peoples to emerging pathogens(6,11-12).
In 2020, American Indians and Alaska Natives were 3.5 times more likely to be diagnosed with COVID-19 than non-Hispanic Whites, and their mortality rate was nearly twice as high(3-4).
COVID-19 presents a significant challenge for Indigenous communities due to social inequalities, limited access to healthcare services, and historical patterns of discrimination, all of which contribute to amplifying the negative impacts of the disease(5). Additionally, Indigenous peoples are more susceptible to respiratory tract infections, and cultural and behavioral factors — such as sharing of drinking gourds and household items, communal housing, and diverse hygiene practices — further facilitate the spread of SARS-CoV-2. These challenges are compounded by the lack of an adequate supply of potable water and limited access to soap and hand sanitizer(6).
Another factor contributing to the poor prognosis of COVID-19 in Indigenous populations is the high prevalence of risk factors within these communities(13-14), such as obesity, hypertension, and diabetes mellitus, which can increase hospitalization rates and mortality(15-16).
Since the heightened risks faced by this vulnerable population and the scarcity of available data, there is a clear need for further research to synthesize and deepen our understanding of the disease's manifestations, symptoms, impacts, and the epidemiological profile of affected Indigenous groups.
The aim of this study is to map the available evidence on COVID-19 in Indigenous populations.
METHOD
Study design
This scoping review protocol will be conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses Extension for Scoping Reviews (PRISMA-ScR)(17) and the methodological guidance of the JBI Manual for Evidence Synthesis(17-18).
One of the main reasons for conducting a scoping review is that, unlike other types of reviews that tend to address more specific questions, scoping reviews aim to map the key concepts within a given research area and to identify existing gaps in literature(18-19).
This protocol was registered on the Open Science Framework (OSF) under registration number 74p58 (available at: https://osf.io/74p58/) to ensure transparency and originality of the review.
The scoping review will follow nine key steps: (a) defining and aligning the objective and the review question; (b) developing and aligning the eligibility criteria; (c) describing the search strategy, data grouping, synthesis, and presentation of results; (d) conducting the literature search; (e) selecting the studies; (f) extracting data; (g) analyzing data; (h) presenting the selected results; and (i) drawing conclusions in line with the proposed objective(17-18).
Review question
The PCC strategy (Population, Concept, and Context) was used to formulate the review question and identify the key concepts(17).
Population
Adults. The World Health Organization (WHO) classifies age groups as follows: child (0–10 years), adolescent (10–19 years), youth (15–24 years), young adult (15–24 years), adult (45–59 years), older adult (60–90 years), and very old (over 90 years)(20). For the purposes of this study, the population will include individuals aged 18 years and older.
Concept
COVID-19. This is a respiratory infection caused by the SARS-CoV-2 coronavirus, which can present symptoms such as fever, fatigue, and dry cough. In some cases, there may also be loss of taste or smell, nasal congestion, conjunctivitis, sore throat, headache, muscle or joint pain, skin rash, nausea, vomiting, chills, and diarrhea(21).
Context
Indigenous. The term “Indigenous” means “native, one who was there before others,” and reflects the cultural and historical diversity of each people(22).
The PCC strategy was used to formulate the following review question: How has COVID-19 manifested in Indigenous populations?
Search strategy
Searches will be conducted in six databases: MEDLINE via PubMed, Cochrane Library, Embase, Web of Science, Scopus, and LILACS. Clinical trial registries such as ClinicalTrials.gov and the World Health Organization International Clinical Trials Registry Platform (WHO ICTRP) will also be consulted. Additional sources will include the British Library (United Kingdom), Google Scholar, and Preprints for Health Sciences (medRxiv), with no restrictions on date or language. The search strategy will use a combination of controlled vocabulary terms — such as Medical Subject Headings (MeSH), Emtree terms and their synonyms, and Health Sciences Descriptors (DeCS) — as well as identified keywords. Boolean operators “AND” and “OR” will be used to combine the descriptors(23-25). The search will be conducted broadly, without the use of database filters, in order to preserve significant samples and minimize the risk of omission. To identify additional studies, manual searches will also be performed in the reference lists of the selected primary studies.
Two independent reviewers will carry out all stages of the scoping review. Based on the PCC strategy, a preliminary search strategy was developed for MEDLINE/PubMed (Figure 1) and will be adapted for use in the other databases.
|
P (Population) |
Adults [>18 years] |
|
#1 |
(Adult[mesh terms] OR Adults[tiab] OR Young Adult[mesh terms] OR Adult, Young[tiab] OR Adults, Young[tiab] OR Young Adults[tiab] OR Middle Aged[mesh terms] OR Middle Age[tiab] OR Aged[mesh terms] OR Elderly[tiab]OR Aged, 80 and over[mesh terms] OR Oldest Old[tiab]) |
|
C (Concept) |
COVID-19 |
|
#2 |
(COVID-19[mesh terms] OR COVID-19[tiab] OR SARS-CoV-2 Infection[tiab] OR Infection, SARS-CoV-2[tiab] OR SARS CoV 2 Infection[tiab] OR SARS-CoV-2 Infections[tiab] OR 2019 Novel Coronavirus Disease[tiab] OR 2019 Novel Coronavirus Infection[tiab] OR 2019-nCoV Disease[tiab] OR 2019 nCoV Disease[tiab] OR 2019-nCoV Diseases[tiab] OR Disease, 2019-nCoV[tiab] OR COVID-19 Virus Infection[tiab] OR COVID 19 Virus Infection[tiab] OR COVID-19 Virus Infections[tiab] OR Infection, COVID-19 Virus[tiab] OR Virus Infection, COVID-19[tiab] OR Coronavirus Disease 2019[tiab] OR Disease 2019, Coronavirus[tiab] OR Coronavirus Disease-19[tiab] OR Coronavirus Disease 19[tiab] OR Severe Acute Respiratory Syndrome Coronavirus 2 Infection[tiab] OR SARS Coronavirus 2 Infection[tiab] OR COVID-19 Virus Disease[tiab] OR COVID 19 Virus Disease[tiab] OR COVID-19 Virus Diseases[tiab] OR Disease, COVID-19 Virus[tiab] OR Virus Disease, COVID-19[tiab] OR 2019-nCoV Infection[tiab] OR 2019 nCoV Infection[tiab] OR 2019-nCoV Infections[tiab] OR Infection, 2019-nCoV[tiab] OR COVID19[tiab] OR COVID-19 Pandemic[tiab] OR COVID 19 Pandemic[tiab] OR Pandemic, COVID-19[tiab] OR COVID-19 Pandemics[tiab] OR SARS-CoV-2[mesh terms] OR SARS Coronavirus 2[tiab] OR Coronavirus 2, SARS[tiab] OR Coronavirus Disease 2019 Virus[tiab] OR 2019 Novel Coronavirus[tiab] OR 2019 Novel Coronaviruses[tiab] OR Coronavirus, 2019 Novel[tiab] OR Novel Coronavirus, 2019[tiab] OR Wuhan Seafood Market Pneumonia Virus[tiab] OR SARS-CoV-2 Virus[tiab] OR SARS CoV 2 Virus[tiab] OR SARS-CoV-2 Viruses[tiab] OR Virus, SARS-CoV-2[tiab] OR 2019-nCoV[tiab] OR COVID-19 Virus[tiab] OR COVID 19 Virus[tiab] OR COVID-19 Viruses[tiab] OR Virus, COVID-19[tiab] OR Wuhan Coronavirus[tiab] OR Coronavirus, Wuhan[tiab] OR COVID19 Virus[tiab] OR COVID19 Viruses[tiab] OR Virus, COVID19[tiab] OR Viruses, COVID19[tiab] OR Severe Acute Respiratory Syndrome Coronavirus 2[tiab]) |
|
C (Context) |
Indigenous people |
|
#3 |
(Indigenous Peoples[mesh terms] OR Indigenous People[tiab] OR People, Indigenous[tiab] OR Peoples, Indigenous[tiab] OR First Nation Peoples[tiab] OR First Nation People[tiab] OR Nation People, First[tiab] OR Nation Peoples, First[tiab] OR People, First Nation[tiab] OR Peoples, First Nation[tiab] OR Native Peoples[tiab] OR Native People[tiab]OR People, Native[tiab] OR Peoples, Native[tiab] OR Natives[tiab] OR Native-Born[tiab] OR Native Born[tiab] OR Indigenous Population[tiab] OR Indigenous Populations[tiab] OR Population, Indigenous[tiab] OR Populations, Indigenous[tiab] OR Indians, South American[mesh terms] OR American Indian, South[tiab] OR Indian, South American[tiab] OR South American Indian[tiab] OR South American Indians[tiab] OR Amerinds, South American[tiab] OR American Amerind, South[tiab] OR Amerind, South American[tiab] OR South American Amerind[tiab] OR South American Amerinds[tiab] OR Indians, North American[mesh terms] OR American Indian, North[tiab] OR American Indians, North[tiab] OR Indian, North American[tiab] OR North American Indian[tiab] OR North American Indians[tiab] OR Amerinds, North American[tiab]OR Amerind, North American[tiab] OR North American Amerind[tiab] OR North American Amerinds[tiab] OR Indians, Central American[mesh terms] OR American Indian, Central[tiab] OR American Indians, Central[tiab] OR Central American Indian[tiab] OR Central American Indians[tiab] OR Indian, Central American[tiab] OR Amerinds, Central American[tiab] OR American Amerind, Central[tiab] OR American Amerinds, Central[tiab] OR Amerind, Central American[tiab] OR Central American Amerind[tiab] OR Central American Amerinds[tiab]) |
|
#4 |
#1 AND #2 AND #3 |
Figure 1 - Search strategy in MEDLINE/PubMed. Vitória, ES, Brazil, 2023
Eligibility criteria
All primary studies, as well as gray literature related to the available evidence on COVID-19 in Indigenous populations, will be included.
Qualitative studies will be excluded, as they do not directly address the objectives of this review, which requires an approach based on aggregated quantitative data, such as morbidity and mortality rates, incidence, prevalence, and geographic distribution of cases.
No language or date restrictions will be applied in the search strategy.
Data extraction
In the first stage, all records retrieved from the six databases will be imported into EndNote software, and duplicate entries will be removed. Two independent reviewers will screen the records by title and abstract using the Rayyan application. In the second stage, full texts of the selected articles will be assessed for eligibility according to the predefined inclusion and exclusion criteria. In cases of disagreement between the two reviewers, a third reviewer will be responsible for making the final decision regarding study inclusion or data extraction.
The study selection process will be outlined using a flowchart, following the PRISMA 2020 model(26) (Figure 2).
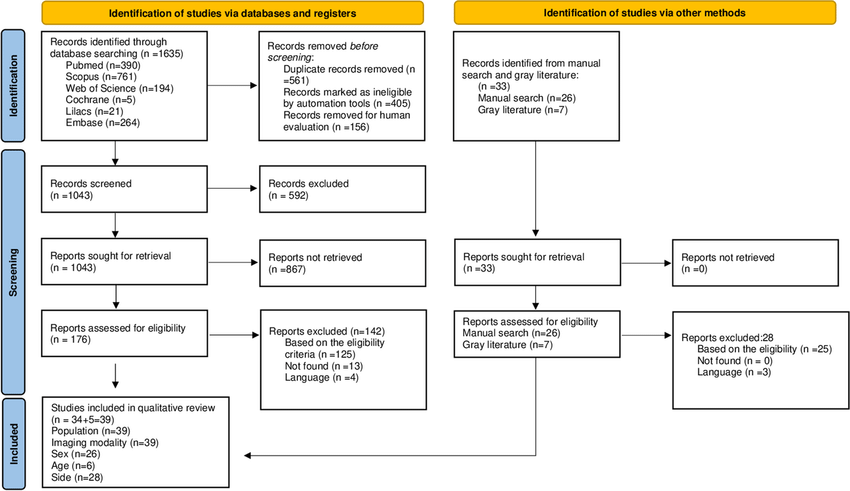
Source: PRISMA flowchart adapted from Page et al., 2021.
Figure 2 - Flowchart of the study selection process. Vitória, ES, Brazil, 2024
The researchers will develop a data extraction tool to organize the information from the selected publications, in accordance with the JBI Manual for Evidence Synthesis. The extracted data will include level of evidence, author(s), journal, year of publication, article title, study objective, study type, country of origin, ethnicity, and main findings.
To present the evidence gathered, visual resources such as graphs and tables will also be used. The most relevant results will be described throughout the manuscript, along with any limitations identified during the review process.
Ethical considerations
This scoping review protocol analyzes only previously published evidence and does not involve human participants. Therefore, ethics committee approval is not required for this type of study.
Expected results
This scoping review will be the first to map the available evidence on COVID-19 in Indigenous populations.
The synthesis of evidence generated may inform the development of public health policies and the implementation of timely preventive measures aimed at reducing the circulation of the SARS-CoV-2 virus in Indigenous territories and mitigating the effects of the COVID-19 pandemic on this highly vulnerable population.
CONFLICT OF INTERESTS
The authors have declared that there is no conflict of interests.
REFERENCES
1. Organização Pan-Americana da Saúde. Histórico da pandemia de COVID-19 [Internet]. Washington: OPAS; 2020 [cited 2023 Jun 12]. Available from: https://www.paho.org/pt/covid19/historico-da-pandemia-covid-19
2. World Health Organization. COVID-19 epidemiological update [Internet]. 169th ed. Geneva: WHO; 2024 [cited 2024 Aug 5]. Available from: https://www.who.int/publications/m/item/covid-19-epidemiological-update-edition-169
3. Huyser KR, Horse AJY, Kuhlemeier AA, Huyser MR. COVID-19 Pandemic and Indigenous Representation in Public Health Data. Am J Public Health. 2021;111(S3):S208-S214. https://doi.org/10.2105/ajph.2021.306415
4. Burki T. COVID-19 among American Indians and Alaska Natives. Lancet Infect Dis. 2021;21(3):325-326. https://doi.org/10.1016/s1473-3099(21)00083-9
5. Cohen JH, Mata-Sánchez ND. Challenges, inequalities and COVID-19: Examples from indigenous Oaxaca, Mexico. Glob Public Health. 2021;16(4):639-649. https://doi.org/10.1080/17441692.2020.1868548
6. Cupertino GA, Cupertino MDC, Gomes AP, Braga LM, Siqueira-Batista R. COVID-19 and Brazilian Indigenous Populations. Am J Trop Med Hyg. 2020;103(2):609-612. https://doi.org/10.4269/ajtmh.20-0563
7. Cunha AA, Nazima MTST, Castilho-Martins EA. Covid-19 among the Brazilian Amazon indigenous people: factors associated with death. Saúde Soc. (Online). 2022;31(2):e210368. https://doi.org/10.1590/S0104-12902022210368en
8. Alves FTA, Prates EJS, Carneiro LHP, Sá ACMGM, Pena ED, Malta DC. Proportional mortality in Brazil’s indigenous population in the years 2000, 2010, and 2018. Saúde em Debate (Online). 2021;45(130):691-706. https://doi.org/10.1590/0103-1104202113010
9. Ministério da Saúde (BR). Saúde indígena: análise da situação de saúde no SasiSUS [Internet]. Brasília: Ministério da Saúde; 2019 [cited 2024 Oct 7]. Available from: https://bvsms.saude.gov.br/bvs/publicacoes/saude_indigena_analise_situacao_sasisus.pdf
10. Marinho GL, Borges GM, Paz EPA, Santos RV. Mortalidade infantil de indígenas e não indígenas nas microrregiões do Brasil. Rev Bras Enferm. 2019;72(1):57-63. https://doi.org/10.1590/0034-7167-2017-0646
11. Cardoso AM, Resende PC, Paixao ES, Tavares FG, Farias YN, Barreto CTG, et al. Investigation of an outbreak of acute respiratory disease in an indigenous village in Brazil: Contribution of Influenza A(H1N1) pdm09 and human respiratory syncytial viruses. PLoS One. 2019;14(7):e0218925. https://doi.org/10.1371/journal.pone.0218925
12. Silva LMVG, Lima BCS, Junqueira TLS. População indígena em tempos de pandemia: reflexões sobre saúde a partir da perspectiva decolonial. Saúde Soc. (Online). 2023;32(2):e220092pt. https://doi.org/10.1590/S0104-12902023220092pt
13. Sardinha DM, Lima KVB, Ferreira AL, Garcez JC, Ueno TM, Rodrigues YC, et al. Clinical and Spatial Characteristics of Severe Acute Respiratory Syndrome by COVID-19 in Indigenous of Brazil. Adv Infect Dis. 2021;11(4):441-454. https://doi.org/10.4236/aid.2021.114039
14. Francisco PMSB, Assumpção D, Bacurau AGM, Leitão VBG, Malta DC. Chronic diseases in the non-village indigenous population: data from the National Health Survey, 2019. Saúde em Debate (Online). 2024;48(1420):e8889. https://doi.org/10.1590/2358-289820241428889I
15. Wu Z, McGoogan JM. Characteristics of and Important Lessons From the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72 314 Cases From the Chinese Center for Disease Control and Prevention. JAMA. 2020;323(13):1239-1242. https://doi.org/10.1001/jama.2020.2648
16. Grasselli G, Zangrillo A, Zanella A, Antonelli M, Cabrini L, Castelli A, et al. Baseline Characteristics and Outcomes of 1591 Patients Infected With SARS-CoV-2 Admitted to ICUs of the Lombardy Region, Italy. JAMA. 2020;323(16):1574-1581. https://doi.org/10.1001/jama.2020.5394
17. Peters MDJ, Marnie C, Tricco AC, Pollock D, Munn Z, Alexander L, et al. Updated methodological guidance for the conduct of scoping reviews. JBI Evid Synth. 2020;18(10):2119-2126. http://doi.org/10.11124/JBIES-20-00167
18. Peters MDJ, Godfrey C, McInerney P, Munn Z, Tricco AC, Khalil H. Chapter 11: Scoping Reviews (2020 version). In: Aromataris E, Munn Z, editors. JBI Manual for Evidence Synthesis. Adelaide: JBI; 2020. https://doi.org/10.46658/JBIMES-20-12
19. Bradbury-Jones C, Aveyard H, Heber OR, Isham L, Taylor J, O’Malley L. Scoping reviews: the PAGER framework for improving the quality of reporting. Int J Soc Res Methodol. 2022;25(4):457-470. https://doi.org/10.1080/13645579.2021.1899596
20. United Nations. World Population Ageing 2020: Highlights [Internet]. New York: United Nations; 2020 [cited 2024 Oct 08]. Available from: https://www.un.org/development/desa/pd/sites/www.un.org.development.desa.pd/files/undesa_pd-2020_world_population_ageing_highlights.pdf
21. World Health Organization. Coronavirus [Internet]. Geneva: WHO; 2024 [cited 2024 Mar 03]. Available from: https://www.who.int/es/health-topics/coronavirus/coronavirus#tab=tab_1
22. Senado Federal (BR). Manual de comunicação da Secom [Internet]. Brasília: Senado Federal; 2024 [cited 2024 Feb 05]. Available from: https://www12.senado.leg.br/manualdecomunicacao
23. Nunes KZ, Grassi J, Lopes AB, Rezende LDA, Cavalcanti JA, Gomes KN, et al. Clinical Indicators of Cardiovascular Risk in Adult Patients Undergoing Chemotherapy: A Protocol for Scoping Review. Pharmacoepidemiology. 2023;2(1):35-41. https://doi.org/10.3390/pharma2010004
24. Lopes-Júnior LC, Siqueira PC, Maciel ELN. School reopening and risks accelerating the COVID-19 pandemic: A systematic review and meta-analysis protocol. PLoS ONE. 2021;16(11):e0260189. https://doi.org/10.1371/journal.pone.0260189
25. Lopes-Júnior LC, Bomfim E, Silveira DSCD, Pessanha RM, Schuab SIPC, Lima RAG. Effectiveness of masstesting for control of COVID-19: a systematic review protocol. BMJ Open. 2020;10(8):e040413. https://doi.org/10.1136/bmjopen-2020-040413
26. Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. Syst Rev. 2021;10:89. https://doi.org/10.1186/s13643-021-01626-4
Submission: 13-Aug-2024
Approved: 02-May-2025
Editors:
Ana Carla Dantas Cavalcanti (ORCID: 0000-0003-3531-4694)
Paula Vanessa Peclat Flores (ORCID: 0000-0002-9726-5229)
Cristiano Gil Regis (ORCID: 0000-0002-6024-6032)
Corresponding author: Carolina Maia Martins Sales (carolina.sales@ufes.br)
Publisher:
Escola de Enfermagem Aurora de Afonso Costa – UFF
Rua Dr. Celestino, 74 – Centro, CEP: 24020-091 – Niterói, RJ, Brazil
Journal email: objn.cme@id.uff.br
|
AUTHORSHIP CONTRIBUTIONS |
|
Project design: Siqueira PC, Sales CMM, Rodrigues JG, Maciel ELN. Data collection: Siqueira PC, Rodrigues JG, Banhos ID. Data analysis and interpretation: Siqueira PC, Banhos ID. Writing and/or critical review of the intellectual content: Siqueira PC, Sales CMM, Rodrigues JG, Banhos ID, Maciel ELN. Final approval of the version to be published: Siqueira PC, Sales CMM, Rodrigues JG, Banhos ID, Maciel ELN. Responsibility for the text in ensuring the accuracy and completeness of any part of the paper: Siqueira PC, Sales CMM, Rodrigues JG, Banhos ID, Maciel ELN. |
