Evaluation of the care cascade in individuals with HIV/AIDS at an ambulatory reference center in Northeast Brazil
Geysa Maria Nogueira Farias1, José Reginaldo Pinto2, Geraldo Bezerra da Silva Junior1, Jeová Keny Baima Colares1, Lara Gurgel Fernandes Távora1, Danielle Malta Lima1
1 University of Fortaleza
2 Ceará State Health Department
ABSTRACT
Background: The continuous care cascade has emerged as a strategy to transpose HIV infection to attain the ultimate goal of care, which is viral suppression. The study aims to describe the care cascade in individuals with HIV / AIDS in a Specialized Ambulatory Service. Methods: The study design is a retrospective cohort of adult individuals with a confirmed diagnosis of HIV/AIDS who initiated their follow-up at the Integrated Medical Care Center (NAMI) of Universidade de Fortaleza (UNIFOR), in the city of Fortaleza, state of Ceará, northeastern Brazil. The following data were used in the investigation: CD4+/CD8+ viral load and HIV viral load. The subjects were classified into two groups: retained (group 1) and non-retained (group 2), totaling a sample of 511 participants.Results: The univariate analysis between these groups showed that there was a significant predominance of male patients with a higher mean age in group 1 (p = 0.01). A history of previous drug addiction and at the first consultation was more frequent in the second group (p = 0.001 and p = 0.01 respectively). Patients from group 1 more often had a history of sexually-transmitted infections (STIs) (p = 0.002) and were more immunosuppressed at the time of diagnosis (p = 0.002). The use of ART at the first consultation was also more significant in the group of retained patients (p <0.001). Conclusions: The study suggests a need to improve the "cascade of care" by expanding it to address dynamic behavioral risks, which can transpose the systemic barriers of access to health services for people living with HIV/AIDS.
Keywords: Acquired Immunodeficiency Syndrome; High-Activity Antiretroviral Therapy; Health Care; Care Cascade.
INTRODUCTION
Despite scientific and technological advances aimed at the treatment and cure of people living with HIV/AIDS (PLHA) and the public policies adopted by several nations and government agencies to fight the disease, it is still a matter of grave concern the number of infected individuals who do not know their serological status, especially in economically developing countries, where infection is concentrated in its epidemic sites [1].
The World Health Organization (WHO) has termed as key populations those defined as the most vulnerable to the transmission and dissemination of HIV/AIDS worldwide, by grouping men who have sex with men, sex workers, transgender individuals, people who use illicit injectable drugs and inmate populations. The Joint United Nations Program on HIV/AIDS (UNAIDS) estimates that 44% of all new HIV infections in adults worldwide occur among key populations and their partners [2,3].
Individuals with HIV who do not adhere to the infection care comprise a heterogeneous group, consisting of those who never reach a certain level of attention, those characterized by a longitudinal process of engagement, disengagement and re-engagement in antiretroviral therapy (ART), as well as those completely unrelated to health care and the care system [4].
Considering these facts, in 2014, the UNAIDS proposed the establishment of goals to maximize the individual and collective benefits related to the diagnosis and treatment of the infection [1]. The continuous care cascade emerged as a strategy to go from the infection to the ultimate goal of care, i.e., viral suppression. This model of care aims to engage and support medical communities to lead efforts to reach, test, treat, and retain key-infection populations in services, as well as the need to address structural barriers – including stigmatization, discrimination, gender violence and, in many cases, criminalization [2].
This cascade structure became known as the 90-90-90 strategy, as it proposes that HIV diagnosis and treatment be expanded by 2020 as follows: 90% of PLHAs will know their serological status; of these, 90% will use ART; and 90% of those undergoing treatment will achieve viral suppression [1,5,6]. In the graphic representation of an HIV care cascade, the left side describes the actions taken for HIV screening and the right side provides information on the efficacy of treatment for those who have been diagnosed [3]. Several cascade models are used worldwide. Some countries use cascades with five bars because they do not have good quality information to differentiate between linked and retained, using only the first of these. On the other hand, others use a seventh bar, “retained in ART”, before the viral suppression bar [5]. The pattern of the Brazilian cascade established by the Ministry of Health consists of six pillars, so divided: PLHA, diagnosed people, linked to care, retained in health care, using ART and with viral suppression [1]. This viral suppression allows the immunological reconstitution, leading to the disease remission in the long term and prolonged survival [7].
In a study that evaluated the cascade of care in several Brazilian states, failures were observed in the prevention of vertical HIV transmission, regardless of the period the infection was diagnosed in the pregnant woman. It was also identified that in the north of Brazil, 20% of pregnant women did not know their HIV status at the time of delivery and 29% had not even been tested during prenatal care. As well as in the northern region, the Brazilian northeast also showed low rates of anti-HIV testing in the prenatal period, respectively, 69.9% and 68.4%. The authors also showed that more than 90% of HIV-positive pregnant women did not use ART during prenatal care, leading to failures in prescribing treatment, even among those who knew they were HIV-positive.
Another identified difficulty was the performance of the elective cesarean-section and the use of ART by the newborn within the first 24 hours of life [8]. In this context, it can be observed that the care cascade response must go beyond the availability of ART, including a timely diagnosis, linkage, retaining and care structuring in the units providing medical care [9].
The aim of this study was to describe the care cascade in individuals with HIV/AIDS in a Specialized Ambulatory Service in the state of Ceará, northeast Brazil.
METHODS
This is a retrospective cohort study of adult individuals with a confirmed diagnosis of HIV/AIDS who initiated follow-up at the Integrated Medical Care Center (SAE NAMI) at Universidade de Fortaleza (UNIFOR), in the city of Fortaleza, state of Ceará, northeastern Brazil. SAE NAMI was implemented in August 2010 and is a specialized outpatient care center for patients with HIV/AIDS.
This study included participants who initiated the follow-up from the implementation of the service in 2010 until December 2017, recorded in the medical files stored in the Medical and Statistical Archive Service (SAME) of the institution.
The study subjects were classified into two groups: individuals who are retained in medical care SAE NAMI or at another service (with at least two HIV viral load tests or two CD4 tests performed in the last year or had ART dispensing in the last 100 days of the year) and individuals who abandoned follow-up – non-retained (did not perform viral load and CD4 tests during the last year or more than 100 days without ART dispensing). The sample consisted of 511 participants.
The diagnosis of HIV infection followed the Ministry of Health Guidelines [10].
In this investigation, the following data obtained from the medical records were analyzed: demographic variables (gender, age, origin and level of schooling); habits (smoking, alcohol consumption, use of illicit drugs in the last three months); behavioral data (sexual partners in the previous year, fixed partner’s serology, sexual orientation, condom use); characterization of HIV infection (classification by the clinical criteria and CDC LTCD4 counts of 1993, occurrence of AIDS-defining diseases); adherence to follow-up (time of follow-up in the service, use of ART, history of ART, treatment adherence); sexually-transmitted infections, laboratory data (first LTCD4 and first viral load (VL), LTCD4 and VL before the first ART, LTCD4 and VL prior to the current ART and the last available LTCD4 and VL exams).
The statistical analysis was carried out using SPSS for Windows, version 23.0 (IBM, USA). Measures of frequency and central tendency were calculated. The Kolmogorov-Smirnov test was used to evaluate the distribution of numerical variables. For the comparative analysis between the two groups, the chi-square test for categorical variables and Student's t or Mann-Whitney test were used for numerical variables, with or without normal distribution respectively. All variables that showed statistical difference with a significance <0.2 in the univariate analysis were included in the logistic regression analysis. For this analysis, p <0.05 was considered statistically significant.
This study is part of a project called CHRONOS that aims to evaluate the epidemiological, clinical and laboratory aspects of HIV/AIDS patients treated at a secondary health unit, after approval by the Ethics and Research Committee of the Universidade de Fortaleza (UNIFOR) under Opinion N. 957,848 (12/12/2015).
RESULTS
This study included 511 participants, of which 83.4% were males, 93.9% were residents of Fortaleza, and 22.5% had a complete or incomplete university/college degree. Regarding the analysis of vulnerabilities, 51.1% reported having multiple sexual partners, 54% were homosexuals, 26.5% had intercourse with HIV-positive individuals. Previous illicit drug use was reported in 27.2% of cases, with marijuana and cocaine being the most frequently used drugs (18.8% and 18% respectively).
More than half of the patients (59.9%) had a previous history of alcoholic consumption and 36.2% had a previous history of tobacco use. In 41.1% of the cases, there was a report of prior sexually transmitted infection or evidenced at the first consultation, with syphilis being the most prevalent one (24.9%), followed by urethritis (8.8%) and condyloma (8.2%) (Table 1).
Table 1. Distribution of the epidemiological clinical profile of patients with HIV/AIDS treated at NAMI, Fortaleza, Ceara, Brazil, 2010 to 2017.
|
Assessed Variables |
Result |
|
Demographic variables |
|
|
Gender Male Female |
426 (83.4%) 85 (16.6%) |
|
Origin Fortaleza Other |
480 (93.9%) 31 (6.1%) |
|
Level of schooling Illiterate Literate Did not finish Elementary School Finished Elementary School Finish/Did not finish High School Finish/Did not finish College/University Unknown |
9 (1.8%) 6 (1.2%) 73 (14.3%) 23 (4.5%) 271 (53%) 115 (22.5%) 14 (2.7%) |
|
Mean age in years (standard deviation) |
33.4 (+/-10.6) |
|
Vulnerability variables |
|
|
Number of partners Fixed Multiple No partner Unknown |
213 (41.7%) 261 (51.1%) 31 (6.1%) 6 (1.2%) |
|
Sexual orientation Homosexual Bisexual Heterosexual Unknown |
276 (54%) 52 (10.2%) 162 (31.7%) 21 (4.1%) |
|
Intercourse with HIV+ individuals |
134 (26.5%) |
|
History of blood transfusion |
24 (4.7%) |
|
Previous illicit drug addiction |
139 (27.2%) |
|
Illicit drug addiction at the time of the 1st Consultation |
71 (13.9%) |
|
Previous history of alcohol consumption |
306 (59.9%) |
|
Alcohol consumption at the time of the 1st Consultation |
265 (51.9%) |
|
Previous history of smoking |
185 (36.2%) |
|
Smoking at the time of the 1st Consultation |
112 (21.9%) |
|
History of STIs |
210 (41.1%) |
|
Number of STIs 1 2 3 |
177 (34.6%) 25 (4.9%) 7 (1.4%) |
|
Clinical variables |
|
|
Presence of comorbidities at the 1st consultation |
286 (56%) |
|
Occurrence of HIV defining diseases at the 1st consultation |
53 (10.4%) |
|
Number of HIV defining diseases 1 2 3 4 |
41 (8%) 9 (1.8%) 3 (0.6%) 1 (0.2%) |
|
CDC Classification according to symptoms at diagnosis: A B C Unknown |
282 (55.2%) 165 (32.3%) 53 (10.4%) 11 (2.2%) |
|
CDC Classification according to CD4 levels at diagnosis 1 2 3 Unknown |
178 (34.8%) 174 (34.1%) 105 (20.5%) 54 (10.6%) |
|
Mean 1st CD4 level performed |
432 cells/dL (+/- 279) |
|
Mean 1st viral load performed |
3.57 log (+/- 1.5) |
|
Use of ART at the first consultation |
257 (50.3%) |
|
CD4 NADIR |
388 cells/dL (+/- 271) |
|
Mean viral load before ART 1 |
4.1log (+/- 1.09) |
|
TOTAL |
511 (100%) |
Clinically, 56% had some comorbidity at the first consultation, with hypertriglyceridemia being the most prevalent one (17%). At the first consultation, 10.4% had a current or previously confirmed diagnosis of some AIDS-defining disease. Among the most frequently identified ones were: esophageal candidiasis, in 20 (26%), pneumocystosis in 15 (19%), extrapulmonary tuberculosis in 13 (18%), and neurotoxoplasmosis in 11 (15%). Regarding the classification by the CDC at the time of the diagnosis, 55.2% belonged to group A (asymptomatic) and 34.8% to group 1 (LTCD4 >500 cells / dL). Approximately half of the patients (50.3%) were on ART at the first consultation and the laboratory parameters showed that the mean of the first LTCD4 test performed was 432 cells/dL (+/- 279), being still slightly above the LTCD4 nadir, which was 288 cells / dL, whereas the first mean HIV viral load was 3.571log (+/- 1.5), as shown in Table 1.
The analysis of retention at the service showed that 63.8% of the patients were undergoing outpatient follow-up and regularly receiving ART dispensed at the pharmacy; 0.2% were undergoing regular follow-up, but ART dispensing at the pharmacy was delayed; 5.7% had abandoned the consultations but were up-to-date with the dispensing of ART at the pharmacy; and 30.3% had fully abandoned treatment. The mean duration of follow-up was 39 months, and 8.4% of the patients had only one consultation. Most patients (71.6%) started ART with regimens containing the combination of 2 nucleoside analogue reverse transcriptase inhibitors and 1 non-analogous one, and 46.8% of patients were still using the first antiretroviral regimen. This association was also the most often prescribed in the ART at the last consultation (65%), at which time 84.7% of the patients were undergoing treatment. Of these, 76.3% reported adherence to ART. The ART response analysis showed that, even after including patients that abandoned treatment, the viral load was below 50 copies /dL (71.5%) and below 1,000 copies/dL (94%) in a considerable number of patients.
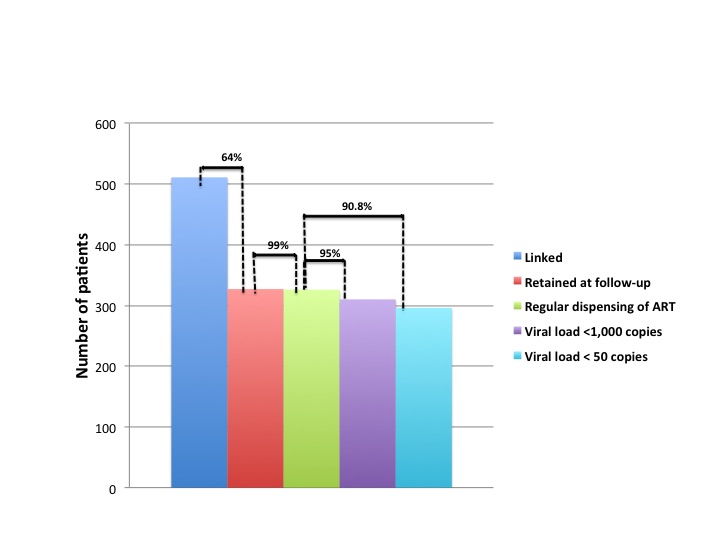
Figure 1 shows the care cascade for patients with HIV / AIDS. It can be observed that, although there is a considerable loss of linkage (64% were still retained at the service), 99% of retained patients are regularly receiving their medications at the pharmacy. Of the latter, 95% had the last VL <1000 copies / dL and 90.8% <50 copies / dL.
Figure 1. Care cascade of patients with HIV/AIDS treated at NAMI, Fortaleza, Ceara, Brazil, 2010-2017.

Figure 2: Flowchart of patients who abandoned treatment for HIV / AIDS in NAMI, Fortaleza, Ceara, Brazil, 2010-2017.
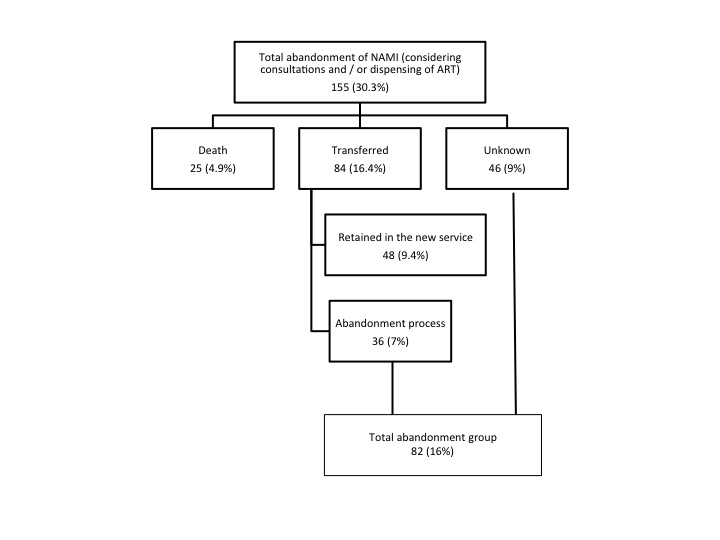
Of the 25 patients who died, only 3 died from non-HIV-related causes: meningococcemia, death by hanging (suicide), and firearm injury.
After the exclusion of these 25 deaths, the patients were then classified into two groups, with group 1 consisting of patients retained at follow-up at SAE NAMI or at another service and group 2 comprising patients who had either fully abandoned treatment or whose follow-up status was unknown (Figure 2). The univariate analysis between these groups showed there was a significant predominance of male patients, with an older mean age in group 1 (p = 0.01). A history of previous drug addiction and at the first consultation was more frequent in the group 2 (p = 0.001 and p = 0.01 respectively). Patients in group 1 more often had a history of STIs (p = 0.002) and were more immunosuppressed at the time of diagnosis (p = 0.002). The use of ART at the first consultation was also more significant in the group of retained patients (p <0.001) (Table 2).
Table 2. Univariate analysis between retained and non-retained HIV / AIDS patients undergoing outpatient follow-up at NAMI, Fortaleza, Ceara, Brazil, 2010-2017.
|
Assessed variables |
Group 1 |
Group 2 |
p |
|
Demographic variables |
|||
|
Gender Male Female |
344 (85.1%) 60 (14.9%) |
61(74.4%) 21 (25.6%) |
0.01 |
|
Level of schooling Illiterate Literate Did not finish Elementary School Finished Elementary School Finish/Did not finish High School Finish/Did not finish College/University Unknown |
6 (1.5%) 3 (0.7%) 54 (13.4%) 18 (4.5%) 212 (52.5%) 99 (24.5%) 12 (3%) |
2 (2.4%) 1 (1.2%) 13 (15.9%) 5 (6.1%) 46 (56.1%) 13 (15.9%) 2 (2.4%) |
0.71 |
|
Mean age in years (standard deviation) |
34 (+/-10.6) |
30.1 (+/-9.0) |
0.01 |
|
Vulnerability variables |
|||
|
Number of partners Fixed Multiple No partner Unknown |
178 (44.1%) 198 (49%) 24 (5.9%) 4 (1.0%) |
32 (39%) 46 (56.1%) 3 (3.7%) 1 (1.2%) |
0.83 |
|
Sexual Orientation Homosexual Bisexual Heterosexual Unknown |
224 (55.4%) 39 (9.7%) 126 (31.2%) 15 (3.7%) |
40 (48.8%) 12 (14.6%) 27 (32.9%) 3 (3.7%) |
0.49 |
|
Intercourse with HIV+ individuals |
106 (26.2%) |
22 (26.8%) |
0.98 |
|
Previous illicit drug addiction |
96 (23.8%) |
34 (41.5%) |
0.001 |
|
Illicit drug addiction at the time of the 1st Consultation |
47 (11.6%) |
18 (22%) |
0.01 |
|
Previous history of alcohol consumption |
246 (60.9%) |
49 (59.8%) |
0.53 |
|
Alcohol consumption at the time of the 1st Consultation |
211 (52.2%) |
46 (56.1%) |
0.06 |
|
Previous history of smoking |
139 (34.4%) |
36 (43.9%) |
0.89 |
|
Smoking at the time of the 1st Consultation |
77 (19.1%) |
28 (34.1%) |
0.27 |
|
History of STIs |
182 (45%) |
22 (26.8%) |
0.002 |
|
Clinical variables |
|||
|
Presence of comorbidities at the 1st consultation |
228 (56.4%) |
41 (50%) |
0.28 |
|
Occurrence of HIV defining diseases at the 1st consultation |
42 (10.4%) |
6 (7.3%) |
0.39 |
|
CDC Classification according to symptoms at diagnosis A B C Unknown |
232 (57.4%) 122 (30.2%) 43 (10.6%) 7 (1.7%) |
40 (48.8%) 33 (40.2%) 6 (7.3%) 3 (3.7%) |
0.25 |
|
CDC Classification according to CD4 at diagnosis 1 2 3 Unknown |
140 (34.7%) 152 (37.6%) 79 (19.6%) 33 (8.2%) |
34 (41.5%) 18 (22%) 14 (17.1%) 16 (19.5%) |
0.002 |
|
Mean 1st CD4 level performed |
431.8 cells/dL (+/- 279) |
486.4 cells/dL (+/- 287) |
0.15 |
|
Mean 1st viral load performed |
3.12 log (+/- 1.5) |
3.0 log (+/- 1.4) |
0.62 |
|
Use of ART at the 1st consultation |
222 (55%) |
25 (30.5%) |
<0.001 |
|
CD4 NADIR |
401 cells/dL (+/- 280) |
346 cells/dL (+/- 214) |
0.38 |
|
Mean viral load before ART 1 |
3.67 log (+/- 1.0) |
3.60 log (+/- 0.96) |
0.49 |
|
TOTAL |
404 |
82 |
|
The logistic regression analysis, however, showed a statistically significant difference between the two groups only regarding the variables previous drug addiction (p = 0.02), history of STIs (p = 0.008), CDC classification by LTCD4 at the time of diagnosis (p = 0.007) and use of ART at the first consultation (p <0.001).
DISCUSSION
Regarding the socio-demographic data, most patients were young male individuals, with high school or higher educational level. Regarding this fact, the last epidemiological bulletin of the Ministry of Health, in the year 2018, showed that 65.2% of the PLHA were in the age group of 20-39 years, 29.4% had finished high school, and most were males [11].
This predominance of HIV infection in male patients can also be seen in other populations. Studies carried out in Turkey and regions outside the sub-Saharan Africa have found similar results. These researchers believe that the higher incidence in young men is due, in part, to the high-risk behavior in terms of transmission, such as contact with prostitutes and homosexuality [12,13].
The evaluation of habits, in this study, showed that most individuals were homosexuals, followed by heterosexuals; a significant percentage of drug users and a history of alcohol addiction and STIs.
The key population at high risk of contracting HIV has progressed from drug users and sex workers to the general public. HIV has become a serious threat to public health and an important limiting factor for social stability and economic development [14].
It is estimated that more than one million individuals are currently living with HIV in the US and many of them have a chronic alcohol addiction. Rates of alcohol abuse are reported by 40 to 50% of the assessed patients. Alcohol and illicit drug use are significant predictors of nonadherence to ART. PLHA who have disorders caused by alcohol and drug use are consistently considered non-adherent to ART and have a higher probability of virological non-suppression and adverse disease outcomes. As HIV infection has become a disease with a more chronic course, comorbid conditions can alter the course of the disease, affecting metabolic function. The use and abuse of illicit drugs is strongly discouraged in this patient population [15,16]. Alcohol use in patients with HIV can cause immunological senescence (the effects of aging on the immune system function), inflammation and hypercoagulability [16,17]. Such a high prevalence in this population is a serious and curious question, since viral infection and alcohol consumption obviously interact in some way to increase their harmful effects [18]. Alcohol use has a strong and consistent association with HIV incidence. The associations between alcohol use and sexual risk behaviors have been consistently identified. Alcohol users have stronger intentions to engage in unprotected sex, poorer sexual communication and negotiation skills, and higher levels of sexual arousal and, therefore, of HIV transmission [16].
Men who have sex with men (MSM) are also disproportionately affected by HIV and other sexually-transmitted infections (STIs). Being diagnosed with an STI is among the most consistent and long-lasting risk factors associated with HIV acquisition. The high risk of HIV acquisition among MSM with bacterial STIs reflects some combination of increased susceptibility, sustained risk behaviors, and sexual network factors [19].
The association between HIV acquisition and STIs is a complex and multidirectional one. Some evidence suggests that genital tract inflammation caused by STIs increases the risk of HIV transmission by increasing HIV-infected partners and due to injury to the genital tract epithelium and recruitment of target cells to this area in partners susceptible to HIV. Rectal infections are direct markers of unsafe receptive anal sex, which is associated with a higher risk of HIV acquisition. Also, because the syphilis epidemic in the US and other developed countries is concentrated among MSM living with HIV, early syphilis can be a marker of unprotected sex within sexual networks, including high-risk MSM living with HIV [19,20].
More than half of the sample in the present study had some comorbidity, but only a small portion had some AIDS-defining disease.
Comorbidities can complicate and accelerate the HIV disease process, manifesting as frailty, organ and function impairment, and increased likelihood of hospitalization and death. The increased prevalence of comorbidities may affect the clinical management of HIV/AIDS patients. These comorbidities may interfere with ART initiation, interrupt ART metabolism, or require drug therapy that may interact with ART, complicating treatment and survival of HIV/AIDS patients. Comorbidities such as coronary artery disease, diabetes mellitus, hypertension, dyslipidemia, bone, liver and kidney disease, chronic respiratory disorders, cancer, and psychiatric and neurocognitive conditions are examples of severe complications that interfere with the infection treatment [21,22,23].
A study carried out in Malaysia has shown that the comorbidities most commonly found in HIV / AIDS patients were infectious diseases, such as hepatitis C, tuberculosis, herpes simplex, oral candidiasis and other opportunistic infections. Tuberculosis coinfections can worsen the quality of life of HIV-infected patients [23].
Studies carried out in other developing countries have reported high mortality rates in hospitalized patients with HIV, when compared to developed countries (up to 38% versus 2.6%). Mortality is associated with advanced stages of the disease, severity of immunological impairment (low CD4 count), presence of opportunistic infections, and lack of resources for care [24].
In our sample, most of the death cases were still related to the evolution of HIV infection. Historically, the development of ART dramatically reduced hospitalization costs, reducing PLHA hospitalization rates. Hospitalized PLHA are now more likely to die from diseases other than AIDS [22,23]. Differences in access to programs, provision of medication and a timely diagnosis have shown to influence patient outcomes [24].
Although more than 60% of the patients had a good follow-up and were retained in the service, a significant percentage (more than 30%) were lost to follow-up at SAE NAMI. If one considers that 48 patients (9.4%) were being followed at another service, one can say that the follow-up retention rate was 73.4%, which is still below the goal recommended by the World Health Organization of 90%. Still, it is a matter of concern that 16% of the cases were classified as full abandonment of follow-up.
Studies carried out in Brazil indicate alcohol consumption, illicit drug use, low family income, low educational level, the presence of psychiatric diseases and a 1- to 3-year interval between the diagnosis of HIV positivity and the start of ART as factors related to treatment nonadherence [25]. Previous studies have identified beliefs about HIV, the stigma associated with HIV status disclosure, food and transportation costs, substance use, pill load, adverse drug events, and lack of communication with health professionals as factors associated with nonadherence to ART [26].
Although we reached percentages >90% at all cascade ranges, except for the change from the first to the second one, it is clear that we still have to improve the retention of these patients. Improvements in cascade indicators over time are probably due to a combination of factors, such as improved access to care and access to ART, availability of treatment regimens that are more effective and easier to follow, changes in guidelines, recommending early onset of treatment and interruptions opposed to treatment, the success of care and treatment initiatives and / or changes in the diagnosed population over time [4,6,27].
The analysis of factors associated with retention or non-retention at the follow-up in NAMI showed that those who maintained adequate follow-up were older (mean of 34 years x 30 years), with lower frequency of drug addiction (23 x 41%), higher frequency of STIs (45 x 26%) and higher frequency of ART use at the 1st consultation (55 x 30%).
Regarding age, it may be presumed that older patients show greater commitment to their treatment or even have caregivers, which somewhat increases adherence to treatment. Drug addiction is a known risk factor for nonadherence to treatment in several clinical settings [15]. The presence of STIs may be a factor that leads the patient to seek the health service and to remain in follow-up due to the presence of symptoms; that is, asymptomatic patients are possibly more careless with their treatment or may not care much about the fact of being HIV-positive, not realizing the severity of the infection. The use of ART at the first consultation may lead to follow-up adherence, as the patients receive full attention from the multidisciplinary team, giving them a sense of being cared for and embraced by the service, making them want to return for the subsequent consultations.
Despite the availability of effective ART, many PLHA do not achieve viral suppression, contributing to AIDS and non-AIDS morbidity and mortality, and ongoing HIV transmission. There is limited evidence-based interventions to improve care retention. More targeted interventions can improve outcomes in the highly vulnerable populations [28].
Sex education in vulnerable groups is also another strategy to change the care cascade among patients in the outpatient treatment facility. Greater challenges in negotiating condom use and new approaches to risk reduction seem to favor significant changes in the quality of life of HIV patients [29].
An important study limitation is associated with the fact that patient sample was concentrated mainly in Fortaleza, and it cannot be verified whether socio-demographic, cultural and behavioral data are similar to those of other regions of the assessed geographic space. Comparisons of different points of care, whether public or private, in Ceará, would be necessary to verify whether the study results would be similar, since the care cascade is still not completely distributed in the countryside of the state, so that the population can have equal access to the services, as in the proposed research scenario.
CONCLUSIONS
This study suggests the need to improve the “care cascade” by expanding it to address dynamic behavioral risks that can transpose the systemic barriers of access to PLHA, expand the service network, and improve patient follow-up retention capacity.
An integrated care cascade that incorporates evolving life experiences and seeks to overcome psychosocial and community-level challenges, aligned with a combined model of health prevention and promotion can more effectively improve the sexual and psychological health of the patients.
ACKNOWLEDGEMENTS
We would like to thank Núcleo de Atenção Médica Integrada (NAMI) of UNIFOR, Ceará, Brazil, for their excellent work caring for individuals living with HIV/AIDS and the availability of official data on the treatment of these patients for this study.
REFERENCES
Miranda WA et al. Modelo preditivo de retenção no cuidado especializado em HIV/aids. Cad Saúde Pública. 2018; 34(10): e00209416.
Wolf RC, Bingham T, Millett G, Wilcher R. Building the evidence base to optimize the impact of key population programming across the HIV cascade. J Int AIDS Soc. 2018 Jul;21 Suppl 5:e25146.
Kowalska JD, Ankiersztejn-Bartczak M, Shepherd L, Mocroft A.. Cascade of care and factors associated with virological suppression among HIV-positive persons linked to care in the Test and Keep in Care (TAK) project. Infection. 2018 Aug;46(4):533-540.
Krebs E et al. Informing targeted interventions to optimize the cascade of HIV care using cluster analyses of health resource use among people living with HIV/AIDS. AIDS Behav. 2018 Jan;22(1):234-244.
Brasil. Ministério da Saúde. Secretaria de Vigilância em Saúde. Departamento de Vigilância, Prevenção e Controle das Infecções Sexualmente Transmissíveis, do HIV/Aids e das Hepatites Virais. Manual Técnico de Elaboração da Cascata de Cuidado Contínuo do HIV. Brasília: Ministério da Saúde, 2017.
Wilton J et al. Trends in HIV care cascade engagement among diagnosed people living with HIV in Ontario, Canada: A retrospective, population-based cohort study. PLoS One 2019 Jan 4;14(1):e0210096.
Nosyk B et al. The cascade of HIV care in British Columbia, Canada, 1996– 2011: a population-based retrospective cohort study. Lancet Infect Dis. 2014 Jan;14(1):40-49.
Miranda AE et al. Avaliação da cascata de cuidado na prevenção da transmissão vertical do HIV no Brasil. Cad. Saúde Pública. 2016; 32( 9 ): e00118215.
Vourli G et al. HIV cascade of care in Greece: Useful insights from additional stages. PLoS One. 2018 Nov 15;13(11):e0207355.
Brasil. Ministério da Saúde. Secretaria de Vigilância em Saúde. Coordenação-Geral da Epidemiologia em Serviços. Guia de vigilância em saúde: volume único. 2ª ed. Brasília: Ministério da Saúde, 2017.
Brasil. Ministério da Saúde. Secretaria de Vigilância em Saúde. Departamento de Vigilância, Prevenção e Controle das Infecções Sexualmente Transmissíveis, do HIV/Aids e das Hepatites Virais (DIAHV). Boletim Epidemiológico HIV/AIDS 2018. Brasília, Ministério da Saúde, v. 49, n. 53, 2018.
Ertunc B, Kaya S, Koksal I. Clinico-Epidemiological Analysis of HIV/AIDS Patients. Eurasian J Med. 2016 Oct;48(3):157-161.
Oliveira RC et al. Scientific Production about the Adherence to Antiretroviral Therapy. Int Arch Med. 2017;10:244. doi: 10.3823/2514
Li JZ et al. The Size of the Expressed HIV Reservoir Predicts Timing of Viral Rebound after Treatment Interruption. AIDS. 2016 Jan 28;30(3):343-53.
Molina PE, Amedee AM, Winsauer P, Nelson S, Bagby G, Simon L. Behavioral, Metabolic, and Immune Consequences of Chronic Alcohol or Cannabinoids on HIV/AIDs: Studies in the Non-Human Primate SIV Model. J Neuroimmune Pharmacol. 2015 Jun;10(2):217-32.
Williams EC et al. Alcohol Use and Human Immunodeficiency Virus (HIV) Infection: Current Knowledge, Implications, and Future Directions. Alcohol Clin Exp Res. 2016 Oct;40(10):2056-2072.
Thangavel S et al. Oxidative Stress in HIV Infection and Alcohol Use: Role of Redox Signals in Modulation of Lipid Rafts and ATP-Binding Cassette Transporters. Antioxid Redox Signal 2018 Feb 1;28(4):324-337.
WingO T, Nesil T, Chang SL, Li MD. Interactive Effects of Ethanol and HIV-1 Proteins on Novelty-Seeking Behaviors and Addiction-Related Gene Expression. Alcohol Clin Exp Res 2016 Oct;40(10):2102-2113.
Katz DA, Dombrowski JC, Bell TR, Kerani RP, Golden MR. HIV Incidence among Men Who Have Sex with Men Following Diagnosis with Sexually Transmitted Infections. Sex Transm Dis. 2016 Apr;43(4):249-54.
Kahle EM et al. Sexually Transmitted Disease Testing and Uptake of Human Papillomavirus Vaccine in a Large Online Survey of US Men Who Have Sex With Men at Risk for HIV Infection, 2012. Sex Transm Dis. 2017 Jan;44(1):62-66..
Abara WE, Smith L, Zhang S, Fairchild AJ, Heiman HJ, Rust G. The influence of race and comorbidity on the timely initiation of antiretroviral therapy among older persons living with HIV/AIDS. Am J Public Health. 2014 Nov;104(11):e135-41.
Zingmond DS, Arfer KB, Gildner JL, Leibowitz AA. The cost of comorbidities in treatment for HIV/AIDS in California. PLoS One. 2017 Dec 14;12(12):e0189392.
Nlooto M. Comorbidities of HIV infection and health care seeking behavior among HIV infected patients attending public sector healthcare facilities in KwaZulu-Natal: A cross sectional study. PLoS One. 2017; 12(2): e0170983.
Álvarez Barreneche MF et al. Hospitalization causes and outcomes in HIV patients in the late antiretroviral era in Colombia. AIDS Res Ther 2017 Nov 13;14(1):60.
Silva JAG et al. Fatores associados à não adesão aos antirretrovirais em adultos com AIDS nos seis primeiros meses da terapia em Salvador, Bahia, Brasil. Cad. Saúde Pública. 2015 ; 31(6): 1188-1198.
Webb Mazinyo E et al. Adherence to Concurrent Tuberculosis Treatment and Antiretroviral Treatment among Co-Infected Persons in South Africa, 2008–2010. PLoS One. 2016 Jul 21;11(7):e0159317.
Auld AF et al. Retention Throughout the HIV Care and Treatment Cascade: From Diagnosis to Antiretroviral Treatment of Adults and Children Living with HIV-Haiti, 1985–2015. Am J Trop Med Hyg. 2017 Oct;97(4_Suppl):57-70.
Meade CM, Badell M, Hackett S, et al. HIV Care Continuum among Postpartum Women Living with HIV in Atlanta. Infect Dis Obstet Gynecol. 2019;e8161495. https://doi.org/10.1155/2019/8161495
Newman PA, Guta A, Lacombe-Duncan A, Tepjan S. Clinical exigencies, psychosocial realities: negotiating HIV pre-exposure prophylaxis beyond the cascade among gay, bisexual and other men who have sex with men in Canada. J Int AIDS Soc. 2018 Nov;21(11):e25211.
Received: 08/28/2019
Revised:05/08/2020
Approved: 05/08/2020